¶ A Layered Approach to Secure the Safety and Graft-Host Integration of hiPSC-derived Functionalized Neuronal Tissueoid for Repair after Complete Spinal Cord lnjury in Rats
Qing-shuai Wei1,5,6, Yuan-huan Ma5,8, Ke-jun Zhang1,2, Wen-tao , Hui-juan , Li-zhi Peng5,7, Jun-hua Wang5,7, Qi-xing , Hou-ge , Ying Liu1,2, Bi-qin Lai5,7, Ge , Ying Ding5,7, Yuan-shan Zeng , Xiang Zeng1,2,3,4,5,7*
-
Lab of Stem Cell Biology and Innovative Research of Chinese Medicine, Guangdong Provincial Hospital of Chinese Medicine/Guangdong Academy of Chinese Medicine/The Second Affiliated Hospital of Guangzhou University of Chinese Medicine, Guangzhou 510120, China
-
National Institute for Stem Cell Clinical Research, Guangdong Provincial Hospital of Chinese Medicine/Guangdong Academy of Chinese Medicine/The Second Affiliated Hospital of Guangzhou University of Chinese Medicine, Guangzhou 510120, China
-
State Key Laboratory of Dampness Syndrome of Chinese Medicine, Guangdong Provincial Hospital of Chinese Medicine, Guangzhou 510120, China
-
Guangdong Provincial Key Laboratory of Chinese Medicine for Prevention and Treatment of Refractory Chronic Diseases, Guangzhou, 510120, China
-
Center for Stem Cell Biology and Tissue Engineering, Key Laboratory for Stem Cells and Tissue Engineering, Ministry of Education, Sun Yat-Sen University, Guangzhou, China
-
College of Basic Medicine and Forensic Medicine, Henan University of Science and Technology, Luoyang,471023, China.
-
Department of Histology and Embryology, Zhongshan School of Medicine, Sun Yat-sen University, Guangzhou, 510080, China
-
Guangzhou Institute of Clinical Medicine, Guangzhou First People’s Hospital, South China University of Technology, Guangzhou, 510180, China.
-
Guangdong Provincial Key Laboratory of Pathogenesis, Targeted Prevention and Treatment of Heart Disease, Guangdong Provincial People’s Hospital(Guangdong Academy of Medical Sciences), Southern Medical University, Guangzhou, 510100, China
* Corresponding Author: Xiang Zeng National Institute of Stem Cell Clinical Research, The Second Affiliated Hospital of Guangzhou University of Chinese Medicine, #55, Nei Huan Xi Road, Guangzhou Higher Education Mega Center, Guangzhou, 510006, China Email: zengxiang@gzucm.edu.cn
* Co-corresponding Author: Yuan-shan Zeng Histology and Embryology, Zhongshan School of Medicine, Sun Yat-sen University, #74, Zhongshan Road, Guangzhou, 510080, China Email: zengysh@mail.sysu.edu.cn
¶ Abstract
The functional integration of donor neurons with host neural pathways is a critical treatment for spinal cord injury (SCl). We constructed human induced pluripotent stem cell (hiPSC)-derived neuronal tissueoids (Ntoids) transfected with thymidine kinase (TK) to transplant donor neurons into SCl rats. Ntoids were produced after 28-days culture in a 3-dimensional gelatin sponge scaffold and possessed neuronal cells with synaptic transmission and neural network property in vitro. Ganciclovir, a TK substrate, depleted proliferative cells in Ntoids without affecting neuronal tissueoid formation, leaving non-mitotic and functionalized neuronal cells for transplantation. Ntoid-treated SCl rats achieved significantly better functional recovery in paralyzed hindlimbs and improved electrophysiological performance. Eight weeks after transplantation, remarkable surviving of donor cells suffused the entire injury/graft site and did not present discernible long-distance cell migration, leading to intensive innervation of the injury/graft site. Ntoids possessed cholinergic hiPSC-derived neurons that survived at the injury/graft site, successfully integrating with regenerating nerve fibers and neurons via synapse-like contacts. Some donor cells differentiated into astrocytes and formed a chimeric blood/spinal cord barrier with the host’s vascular epithelium. Ntoids represent a structural and functional neuronal relay to restore functionality in paralyzed limbs. These findings support the safety and efficacy of the transplantation of mature neuronal tissue with safety-regulatory mechanisms to treat SCI and provide a framework for future clinical application using tissue-engineered Ntoid.
Keywords: Human induced pluripotent stem cells, Neuronal tissueoid, Transplantation, Motor function recovery, Spinal cord injury.
¶ 1. Introduction
In adult mammals, spinal cord injury (SCI) leads to irreversible tissue loss, including neurons and glia[1], resulting in motor, sensory and autonomic function deficits below the level of injury[2]. The limited spontaneous regeneration[3] and harsh microenvironment[4] caused by secondary injury makes it difficult for endogenous regeneration to compensate lost tissue in adult mammals. The exogenous neural grafts, including stem cell therapy and tissue engineering technology, may offer a promising therapeutic strategy to reconstruct neural pathways after SCI[5,6]. Recently, inspirational advancements in research have revealed promising therapeutic capacities, such as neural protection[5,7], immune modulation[8], and neuronal replacement[9], following the grafting of exogenous neural stem/precursor cells. However, safety issues caused by unidirectional migration[10], ectopic neurogenesis[11] and the uncertain differentiation[12] of donor cells impedes the clinical application of this regimen. To augment the retention of donor cells and subsequent neuronal differentiation, significant modification of the injury microenvironment is required[13,14].
In our previous study, we used tissue engineering methodology to construct stem cell-derived neural network tissue to improve safety issues and eliminate the uncertain differentiation of donor cells[15]. Following transplantation of neural network tissue derived from either hippocampus-derived neural stem cells (NSCs) or bone marrow-derived mesenchymal cells (MSCs) into the injury site in a rat model, surviving donor cells were observed at the injury/graft site for eight weeks and participated in the reconstruction of neural pathways without ectopic migration following SCI. More importantly, the voluntary motor function of paralyzed hindlimbs was significantly improved following transplantation[16,17]. Furthermore, the safety and efficacy of transplanted NSC- or MSC- derived neural network tissue was successfully demonstrated in canine models[18,19] with large defects in the spinal cord tissue. Collectively, these results suggested that the transplantation of stem cell-derived neurons generated by a tissue engineering approach is a promising strategy with which to repair SCI.
However, the source of cells in our previous study was limited to adult multipotent stem cells. The safety and efficacy of embryonic/pluripotent stem cell-derived neurons in the treatment of SCI treatment has yet to be fully investigated. Induced pluripotent stem cells (iPSCs), due to their ease of acquisition, lack of ethical restriction, and multidirectional differentiation potential[20–22] may serve as an attractive alternative cell source for human neural repair. Accumulating recent evidence suggests that transplanted human iPSC (hiPSC)-derived neural progenitor cells (hNPCs) can integrate with neural pathways in the host and promote the recovery of voluntary motor function[23,24]. However, the long time period required for full neuronal maturation[25,26] may compromise the therapeutic efficacy[25] of this approach as human NSCs or hNPCs may retain an intrinsic human rate of maturation[27].
In view of this, it would be desirable to test whether the transplantation of hiPSC-derived neurons may yield earlier functional restoration in SCI. Due to the presence of undifferentiated pluripotent cells in the graft, and to reduce the risk of teratoma formation after transplantation, it is necessary to establish an efficient in vitro induction system with strict screening procedures to eliminate tumorigenicity. Therefore, in the present study, we adopted a stepwise approach to secure safety and graft-host integration, including a safe cell system based on suicide genes, the stepwise induction of neuronal differentiation, tisssueoid culture and cells seeded in a biological scaffold. Analysis revealed that donor cells survived well in the spinal cord injury/graft site, with no ectopic migration or teratoma formation. Furthermore, the grafted neurons integrated with host neural pathways and acted as a “neuronal relay” to repair the motor functionality of paralyzed hindlimbs in rats with complete SCI.
¶ 2. Methods
¶ 2.1. Ethics statement
All animal experiments were reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) of Sun Yat-sen University (Approval number: SYSU-IACUC-2018-000273) and the Affiliated Hospital of Guangzhou University of Chinese Medicine (Approval number: 2023119). Animal welfare protocols adhered strictly to the Laboratory Animal Guidelines for Ethical Review of Animal Welfare issued jointly by the General Administration of Quality Supervision, Inspection and Quarantine of the People’s Republic of China and the Standardization Administration of China. All efforts were made to minimize animal suffering and reduce sample size.
¶ 2.2. Maintenance of the hiPSC line
The hiPSC line was provided by the Key Laboratory for Stem Cells and Tissue Engineering, Ministry of Education, Sun Yat-sen University[28], and was maintained on Matrigel® (Corning Incorporated, Corning, New York, United States)-coated 6-well plates using mTESRTM1 medium (STEMCELL Technologies Inc., Vancouver, British Columbia, Canada) at in an atmosphere containing with daily medium replacement. To identify pluripotency, hiPSCs were fixed with paraformaldehyde in 0.1M phosphate buffer (PB, pH7.4) at room temperature for 1hour. Then, immunofluorescence staining of Nanog, Octamer binding transcription factor 4 (OCT4),
Paired Box 6 (Pax6) and SRY-Box Transcription Factor 1 (Sox1) was performed on hiPSCs that were negative for mycoplasma contamination, as verified using MycAway \textsuperscript { \textregistered } Mycoplasma Real-time qPCR Detection Kit(2G) (Yeasen Biotechnology (Shanghai) Co., Ltd., Shanghai, China).
¶ 2.3. Induction of hiPSC-derived human neural progenitor cells/neurons
hiPSCs were differentiated into human neural progenitor cells (hNPCs) using STEMdiff SMADi Neural Induction Medium (STEMCELL Technologies Inc., Vancouver, British Columbia, Canada) in accordance with the manufacturer’s specifications. In brief, hiPSCs were cultured in STEMdiff SMADi Neural Induction Medium on a Matrigel®-coated-6-well plate with daily medium replacement. Neural induction efficiency was confirmed by morphological assessment within 14 days. For immunophenotypic characterization, the derived cells were fixed with paraformaldehyde in 0.1M PB (pH7.4) at 24 hours or 14 days, followed by immunofluorescence staining with antibodies against hNestin, Sox1, and Nanog (a negative selection marker). Expanded hNPCs were subsequently maintained in Neural Progenitor medium (STEMCELL Technologies Inc., Vancouver, British Columbia, Canada) with daily medium changes.
Neuronal differentiation was performed using neuron induction medium, including (V/V) DMEM/F12 (Gibco (Thermo Fisher Scientific), Grand Island, New York, United States), (V/V) NeurobasalTM-A(Gibco, Grand Island, New York, United States), (V/V) N-2 Supplement (Gibco, Grand Island, New York, United States), (V/V) B-27 Supplement (Gibco, Grand Island, New York, United States), BDNF (R&D Systems, Inc. (a Bio-Techne brand), Minneapolis, Minnesota, United States) and db-cAM P(Millipore Sigma, Burlington, Massachusetts, United States) to induce hNPCs to become neurons, as previously described[29]. In brief, hNPCs were cultured in neuron induction medium on a laminin-coated-24-well plate, and the medium was changed every other day. After 14 days, the hNPCs-derived cells were fixed with paraformaldehyde in 0.1M PB for immunofluorescence staining with antibodies against microtubule associated protein 2 (MAP2), neurofilament 200 (NF200), neuronal nuclei (NeuN), beta-tubulin III (Tuj1), growth associated protein 43 (GAP43) and glia fibrillary acidic protein (GFAP).
¶ 2.4. Generation of GFP-TK expressing human neural progenitor cells (hNPCs-GFP-TK)
The hiPSC line was transduced with recombinant lentiviral vectors encoding enhanced GFPthymidine kinase (TK) under the EF1α promoter (Len-EF1 -GFP-TK) at a multiplicity of infection (MOI) of 1. Transduction was performed in mTeSR™1 medium supplemented with polybrene (Millipore Sigma, Burlington, Massachusetts, United States) and Y-27632 dihydrochloride (Tocris Bioscience, Bristol, United Kingdom) for 24–48h. Following viral transduction, GFP-TK expression was confirmed by fluorescence microscopy prior to selection with blasticidin S HCl (Gibco, Grand Island, New York, United States) until the proportion of GFP-TK expressing hiPSCs (hiPSCs-GFP-TK) was . GFP-TK expressing populations were subsequently enriched by fluorescence-activated cell sorting with excitation/emission at using flow cytometry (FACS).
FACS-purified hiPSCs-GFP-TK were expanded before initiating neural differentiation, as described in Section 2.3. At 14 days post-induction, immunofluorescence analysis was performed using antibodies against definitive neural progenitor markers (Sox1, Pax6 and hNestin).
¶ 2.5. Use of the TK-GCV system to eliminate proliferative cells
Next, we quantitatively evaluated the cytotoxicity of ganciclovir (GCV) and the efficacy of the TK-GCV system with regards to eliminating proliferating cells. hiPSCs-GFP-TK, hNPCs-GFP-TK, wild-type hiPSCs, and wild-type hNPCs were seeded at equivalent densities in Matrigel® (Corning Incorporated, Corning, New York, United States)-coated 24-well plates. After 24 hours of cell adhesion, graded concentrations of ganciclovir (GCV: 0, 3, 10, and ) were administered to triplicate wells for each condition. Following seven days of culture under standard conditions , cells were fixed with paraformaldehyde (PFA) and nuclei were counterstained with Hoechst 33342(Millipore Sigma, Burlington, Massachusetts, United States). Five non-overlapping fields per well were systematically imaged using a Zeiss LSM 800 confocal microscope(Carl Zeiss AG, Oberkochen, Germany). Nuclear quantification was performed with ImageJ(National Institutes of Health, Bethesda, Maryland, United States). Cell viability was normalized using the mean cell count of the GCV control group as the baseline (defined as survival). The survival rate at each concentration was calculated using the following formula: Survival Rate .
Next, we established a standard curve to quantitatively assess residual undifferentiated hiPSCs in hNPCs and hiPSC-derived neurons. Serial dilutions of hiPSCs with HaCaT keratinocytes (at ratios of 1:1,000; 1:5,000; 1:10,000; 1:50,000; and 1:100,000) were plated onto Matrige \textsuperscript { \textregistered } (Corning Incorporated, Corning, New York, United States)-coated 24-well plates. Total RNA was then isolated using the FastPure Cell/Tissue Total RNA Isolation Kit (Vazyme Biotech Co., Ltd, Nanjing, China), followed by cDNA synthesis with HiScript III RT SuperMix for qPCR (Vazyme Biotech Co., Ltd, Nanjing, China). Quantitative real-time PCR (qPCR) was then performed using ChamQ Universal SYBR qPCR Master Mix (Vazyme Biotech Co., Ltd, Nanjing, China) on a QuantStudio 5 system (Thermo Fisher Scientific, Grand Island, New York, United States). Following GCV treatment, residual hiPSC contamination was determined by measuring the expression of Teratocarcinoma-Derived Growth Factor 1 (TDGF1, Gene ID: 6997) with the following validated primers: Forward: 5’-TTTGAACTGGGATTAGTTGCCG-3’; Reverse: 5’-GGGGCCAAATGCTGTCATCT-3’. Residual proliferating cells in hiPSCs-GFP-TK-derived neurons as described above. TDGF1 was proven to be more reliable for detecting the proportion of residual pluripotent stem cells, relative to OCT4[30].
¶ 2.6. Construction of hiPSC-derived neuronal tissueoids (Ntoids)
Three-dimensional (3D) gelatin sponge scaffolds (3D-GS) were prepared as previously described[31]. In brief, the cylindrical 3D-GS scaffold ( in diameter and in length) underwent ethylene oxide sterilization. Before cells were seeded to the 3D-GS, the scaffolds were soaked with the neuron induction medium containing laminin for 12 hours.
A total of hNPCs were suspended in neuron induction medium and seeded into the 3D-GS. To optimize cellular adhesion, the 3D-GS with implanted hNPCs was incubated at for in a 24-well plate before adding of neuron induction medium; then, another of neuron induction medium was added after . Cultures were maintained with medium replenishment every 48 hours. After 14–28 days of differentiation, the Ntoids were fixed as described earlier and stained with a range of neuronal markers: MAP2, synapsin I (Syn1), sodium channel protein (NaV), calcium channel protein , choline acetyltransferase (ChAT), glutamate (Glu) and -aminobutyric acid (GABA).
hNPCs-GFP-TK were used for transplantable Ntoid construction. After 14–28 days of 7 differentiation, random scaffold sampling per batch) underwent Tuj1 immunofluorescence quantification to confirm neuronal differentiation efficiency prior to in vivo implantation.
¶ 2.7. Scanning electron microscope (SEM) analysis
SEM was utilized to investigate cellular adhesion dynamics and morphological features within the Ntoids. The Ntoids were triple-washed with 0.1M PB , fixed in 0.1M PB containing glutaraldehyde, for 4h at , triple-washed with 0.1M PB (pH7.4); and dehydrated with a series of ethanol concentrations ( , , , ) with intervals. Dried Ntoids were then coated with gold and observed by SEM (Royal Philips Electronics, Eindhoven, The Netherlands). The Ntoids were imaged under high vacuum mode Pa) using a field-emission SEM at accelerating voltage with working distance maintained at .
¶ 2.8. Transmission electron microscope (TEM) analysis
TEM was used to analyze synaptic connections between hiPSC-derived neurons in the Ntoids. After 14–28 days of differentiation, the Ntoids were triple-washed with 0.1M PB , fixed in 0.1M PB (pH7.4) containing glutaraldehyde for 4 hours at , osmicated with osmic acid for 1 hours at room temperature; and dehydrated with a series of ethanol concentrations , , , ) with intervals. Dried Ntoids were then embedded in Epon overnight at room temperature and polymerized for 48 hours at . Ultra-thin sections were then cut on an ultramicrotome and observed by TEM (Royal Philips Electronics, Eindhoven, The Netherlands).
¶ 2.9. In vitro electrophysiology
Whole-cell patch clamping was used to investigate the functional maturation of hiPSC-derived neurons in the Ntoids. After 28 days of differentiation, an EPC Amplifier 10 and Patchmaster software(HEKA Elektronik, Langen, Germany) were used to collect electrical signals, with a resistance of . The external solution contained 5mM KCl, 140mM NaCl, lmM MgCl, 2mM CaCl, 10mM HEPES and10mM glucose (320mOsm; pH7.3). Electrophysiological recordings of hiPSC-derived neurons in the Ntoids were performed at room temperature. All drugs were applied using a micro-perfusion system. Cells were tested for action potential induction and spontaneous excitatory postsynaptic current (sEpSC) generation. All data were collected and analyzed using Mini Analysis software (Synaptosoft, Leonia, New Jersey, United States).
Field potential and neuroconnectome analysis of Ntoids were conducted using a high-density microelectrode array system (HyperCAM Alpha, 3Brain, Switzerland). The Ntoid was gently transferred from the culture plate to the center of the electrode area on the chip with 3D microelectrodes (CorePlate™ 6W-3D, 3Brain). Finely adjusted the position of the Ntoid to cover as more electrode areas as possible. Gently added of Ntoid culture medium to keep the organoid moist. A specialized sample press net was used for tissue fixation. Each sample was recorded continuously for 5 minutes. After recording, electrophysiological data was analyzed using BrainWave5 (3Brain).
¶ 2.10. Surgery and transplantation
Three days prior to surgery, adult female Sprague-Dawley (SD) rats ( in weight) were given a daily subcutaneous injection of tacrolimus , mycophenolate mofetil and prednisone . All rats were deeply anesthetized with an intraperitoneal injection of sodium pentobarbital with toe-pinch reflex monitoring. T10 complete laminectomy was performed under a surgical microscope to expose a spinal cord segment; then, we used extradural microsurgery to completely resect a segment of spinal cord tissue, including the attached spinal roots. The 14-day Ntoids (Ntoid group, ), hNPC-seeded 3D-GS (hNPCs group, ) or no cell-seeded 3D-GS (GS group, ) were then implanted into the gap in the spinal cord. A pilot study using non-TK-GCV hNPC-seeded 3D-GS ) was also performed. Once hemostasis had been achieved, the overlying musculature and skin were sequentially sutured. The animals received extensive post-operative care, including the intramuscular injection of penicillin per day) for three days and stroke-physiological saline solution ( per day) for 7 days. All animals received manual emiction three times daily until automatic micturition function was re-established. Immunosuppressants were administered daily for 8 weeks, including tacrolimus , mycophenolate mofetil and prednisone .
¶ 2.11. Behavior assessments
All animals were trained to climb onto a inclined grid and freely explore an open field before surgery. The Basso, Beattie, and Bresnahan (BBB) test[32] was performed for three days and every week after surgery. During open-field locomotion testing, all animals were allowed to freely explore an open field for to assess hindlimb locomotor frequency, joint movement range, weight bearing, and coordination. The inclined grid test[33] was performed in the final week to qualitatively assess the accuracy of foot placement and coordination according to what was reported previously by Ramon-Cueto et al.
¶ 2.12. Electrophysiology
Eight weeks after surgery, cortical motor-evoked potentials (CMEPs, were recorded to evaluate the functional connectivity of the reconstructed corticomotor pathway by grafting Ntoids using a Neuro-Exam M-800 Data Acquisition Analysis System(Zhuhai Medcom Technology Co., Ltd., Zhuhai, China). All rats were anesthetized via the inhalation of (v/v) isoflurane in air ( with core temperature maintained at using a homeothermic pad. All rats received Bregma-referenced craniotomy to expose the sensorimotor cortex (SMC). Continuous saline irrigation was used to maintain tissue hydration during drilling. The stimulating electrode was positioned in the sensorimotor cortex (SMC), while the recording electrode was interfaced with the hindlimb sciatic nerve. CMEPs were recorded following electrical stimulation of the SMC, with single-pulse stimulation ( , 50-ms duration) administered five times per animal.
¶ 2.13. Tissue processing
All animals were euthanized eight weeks post-surgery. All animals were deeply anesthetized via the intraperitoneal injection of pentobarbital sodium . Intracardiac perfusion was performed using heparinized saline ( NaNO₂, heparin) followed by paraformaldehyde (PFA) in PBS . The spinal cord was removed and post-fixed in the same fixative for 24 hours, and then placed into sucrose in PB for two days followed by sucrose in PB for 5 days at . Successive T8–T12 successive segments of the spinal cord were embedded in optimal cutting temperature (OCT) compound and longitudinally sectioned into slices using a CryoStar NX50(Thermo Fisher Scientific, Grand Island, New York, United States).
¶ 2.14. Immunofluorescence staining
Immunofluorescence staining was performed as described earlier. In brief, the sections were blocked with goat serum for 1 hours at and then sequentially incubated with primary antibodies overnight) and species-matched secondary antibodies , 1 hours); antibody specifications are provided in Supplementary Table 1. Nuclear counterstaining was performed using Hoechst 33342 (Hoe) prior to mounting. Imaging was acquired on a Zeiss LSM 800 confocal microscope, using consistent parameter settings across all samples.
¶ 2.15. Morphological quantification
Next, we used in vitro experiments to quantitatively evaluate the pluripotency maintenance of hiPSCs and determined maintenance efficiency by comparing pluripotency markers (Nanog+ or ) and hNPCs markers or ) against the total number of cells. To quantitatively evaluate the neural differentiation capacity of hiPSCs in vitro, the differentiation efficiency of hNPCs was quantified by comparing hNPCs markers , or hNestin⁺), neural lineage markers or ) and pluripotency markers (Nanog+ or ) against the total number of cells. To quantitatively evaluate the neuronal differentiation capacity of hNPCs in vitro, the differentiation efficiency of neurons was quantified by comparing neuronal markers , or hNestin⁺), neural lineage markers , , NeuN+ and Tuj1+) and astrocyte markers ) against the total number of cells. All immunostaining experiments included technical triplicates with three biological replicates, and marker specificity was validated by isotype control staining.
To quantitatively evaluate the survival of donor cells in vivo, we quantified GFP fluorescent density by calculating the ratio of pixel area to total injury/graft(I/G) site in spinal cord. In addition, the proportion of human nuclear antigen (hNu) cells were quantified by calculating the number of cells out of the total number of cells. To quantitatively evaluate the neuronal differentiation of donor cells, NF and Tuj1 fluorescent density was quantified by calculating the ratio of or area to the total I/G site. In addition, the proportion of NeuN⁺ cells was quantified by calculating the number of NeuN + cells versus the total number of cells. To quantitatively evaluate axonal projection of the donor cells, the number of processes extending from donor cells (with a length ), were calculated , and
3mm caudal to the injury site. To quantify positivity, one in every five of the whole series of sections from each animal was selected ).
All high-resolution images were acquired using a LSM800 confocal laser scanning microscope. Quantitative image analysis was performed using Adobe Photoshop software(Adobe Systems Incorporated, San Jose, California, United States) thresholding tools with batch processing to ensure consistency.
¶ 2.16. Statistical analysis
All quantitative data were analyzed using the Student’s t-test or by one-way analysis of variance (ANOVA) and reported as means standard deviation and analyzed using SPSS version 22.0 software(IBM Corporation, Armonk, United States). In the case of equal variance, the least-significant difference (LSD) test was applied; otherwise, Dunnett’s T3 post-hoc test was used. The significance level was set at .
¶ 3. Results
¶ 3.1. Stepwise construction of neuronal tissueoid
Neuronal tissueoids derived from HiPSCs were constructed by a neural progenitor cell (NPC) induction phase and a bioscaffold-based neural tissueoid (Ntoid) construction phase (Figure 1A). Following induction, hiPSCs shifted from a pluripotent stage to a neural progenitor stage (Supplementary Figure 1) featuring hNestin (Figure 1B, D), Sox1 (Figure 1C, D), Pax6 (Supplementary Figure 2C, G) and negligible Oct4 (Supplementary Figure 2E, G) after 14 days of two-dimensional (2D) induction. This process yielded high purity hNPCs with robust propagation capability up to 10 passages and potent neuronal differentiation capability. Next, hNPCs were seeded into cell dishes to induce neurons. After 14 days, the cells expressed a high proportion of neuronal markers, such as MAP2 , Supplementary Figure 2H, N), NF200 , Supplementary Figure 2I, N), NeuN , Supplementary Figure 2J, N), Tuj1 , Supplementary Figure 2K, N), and growing nerve fiber marker GAP43 (Supplementary Figure 2M), but a low proportion of the astrocyte marker GFAP ( , Supplementary Figure 2L, N). Next, hNPCs from passage 3–6 (P3-6) were seeded into 3D-GS scaffolds with good cytocompatibility for Ntoid construction. After 14 days, Ntoids expressed a high proportion of neuronal markers, such as NF (Figure 1F) and Tuj1 (Figure 1G), thus suggesting that the Ntoid were predominantly formed from a neuronal population.
To validate the relationship between neurons and neuronal function, we next performed morphological and electrophysiological analysis of Ntoids (Figure 1A). SEM revealed clusters of cell aggregates inside the sponge-like scaffold. The cells in neighboring aggregates were linked by bundles of neurites resembling a network feature (Figure 1J). The bundles of cell neurites and connections increased from 14 days to 28 days in the scaffold (Figure 1J, K). Immunostaining revealed that the cells in the aggregates were predominantly MAP2 immunopositive neurons with an abundance of co-staining with synaptic marker Syn1 (Figure 1L-L1). The appearance of mature synaptic structure was further validated by TEM, which revealed round synaptic vesicles in the presynaptic component and a thickened membrane. Dense postsynaptic areas in the postsynaptic component, a feature of asymmetric excitatory synapse, were also observed (Figure 1M-N1). This synaptic structure belonged to an asymmetric excitatory synapse. The proportions of ChAT (Figure 1H) and Glu (Figure 1I) immunopositive neurons in Ntoids were significantly greater that those of GABA immunopositive neurons (Supplementary Figure 2Q). Furthermore, immunostaining showed that sodium channel protein (NaV, Supplementary Figure 2O) and calcium channel protein (CaV, Supplementary Figure 2P) were both expressed in the neurons of Ntoids.
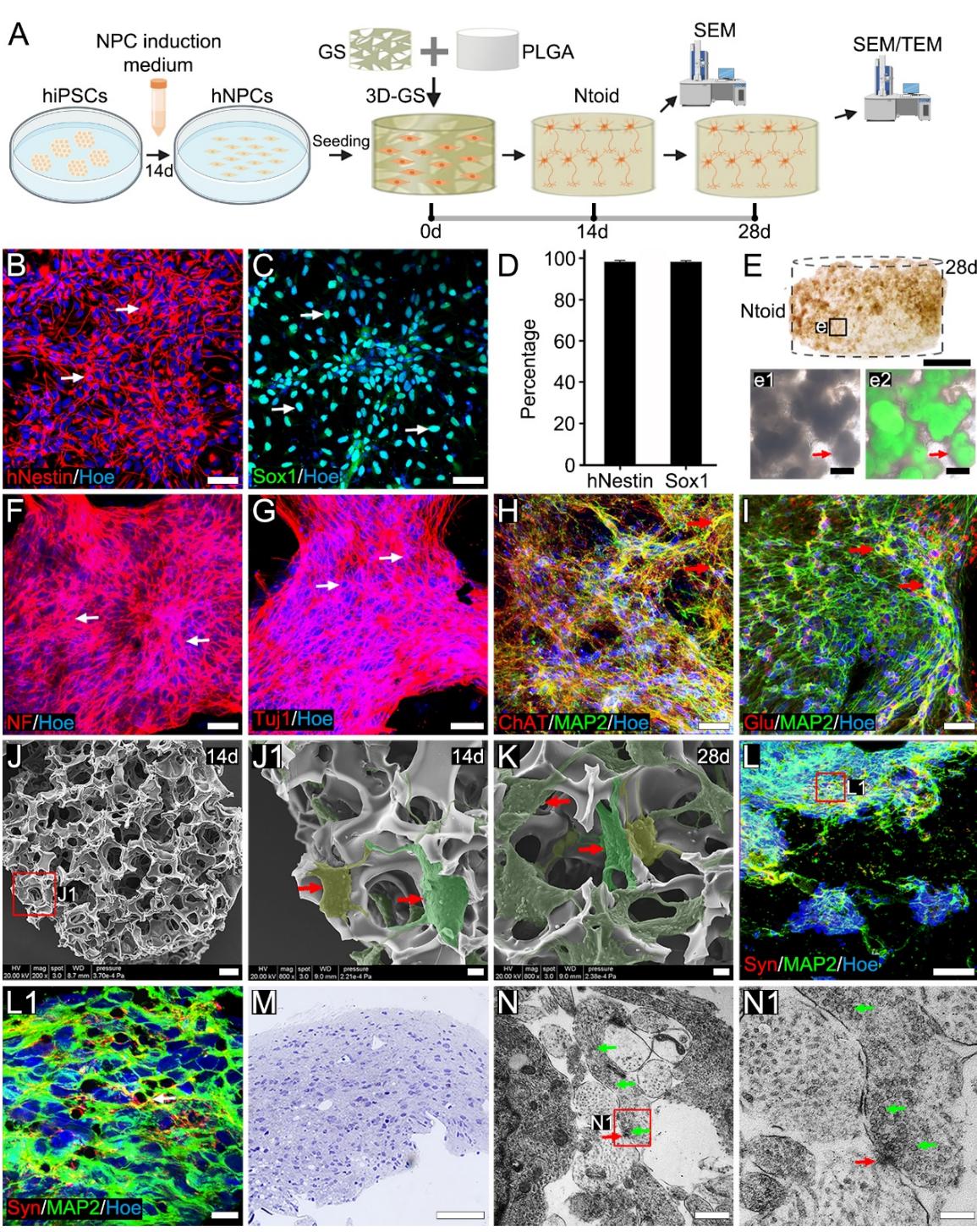
Figure 1. Stepwise construction of a neuronal tissueoid. (A): Schematic diagrams of the stepwise construction of neuronal tissueoids (Ntoids). (B-C): Immunofluorescence staining showing the high expression of hNestin (arrows, B) and Sox1 (arrows, C) in human neural progenitor cell (hNPCs). (D): Bar chart showing the proportions of hNestin+ and cells in Hoe+ cells. (E-e2): The distribution of hiPSC-derived cells in an Ntoid under inverted phase contrast microscopy. (F-G): Immunofluorescence staining showing the high expression of NF (arrows, F) and Tuj1 (arrows, G) in an Ntoid. (H-I): Immunofluorescence staining showing ChAT+/MAP2+ (arrows, H) and Glu+/MAP2+(arrows, I) neurons in an Ntoid. (J-K): SEM showing the morphology of hiPSC-derived cells in an Ntoid after seeding for 14 days (J-J1) or 28 days (K); a higher magnification image (J1) is shown in the boxed area in (J), demonstrating that the clusters of cells were linked by bundles of their neurites (arrows, J1). (L-L1): Immunofluorescence staining showing presynaptic terminals and neurons in an Ntoid (L); a higher magnification image (L1) is shown in the boxed area in (L), demonstrating presynaptic terminals contacting neurons (arrows). (M): Light microscopy showing a semi-thin section of an Ntoid. (N): TEM showing a synapse-like structure (red arrow) at the point of contact between a neurite and vesicles (green arrows). (N1): A higher magnification is shown in the boxed area in , showing a junctional site (red arrow) with electron-dense cell membranes, which were similar to the presynaptic membrane and postsynaptic membrane, and some synaptic vesicles (green arrows) on one side of the cytoplasm at the cell junction. Scale bars (B-C, F-I, L and M), (E), (J and e1-e2), (J1-K), (L1), (N), and (N1).
¶ 3.2. Function maturation and neural network formation of Ntoids
Whole-cell patch clamping was applied to detect mature neuronal function of each selected cell in Ntoid (Figure 2A). Evoked action potentials, an index of electrophysiological finctionality in mature neurons, were recorded in approximately of cells sampled from Ntoids with an extended culture time to 28 days (Figure 2B-B1). In addition to inhibitory postsynaptic currents (IPSCs), excitatory postsynaptic currents (EPSCs, Figure 2C-C1) were also recorded, thus suggesting the fommation ofan excitatory neuronal population within Ntoids. High-density MEA was applied to assess the electrical activity of neuronal populations in Ntoids and the correlations between their activities (Figure 2A). The electrode signal maps showed rich electrical activity recorded by electrodes in the Ntoid-covered area (Figure 2D, E), reflecting active neuronal population activity in Ntoids. Numerous spike signals and typical spike waveforms were observed (Figure 2F, G). The maximum spike amplitude reached . The spike amplitude distribution indicates that amplitude level was peaked around (Figure 2H). Raster plots reveal clusters of burst firing in Ntoids (Figure 2I). The neural network connectivity map, derived from cross-correlation analysis between electrodes, shows signal-sender neurons, signal-receiver neurons, and signal-broker neurons distributed within the Ntoid, forming an interconnected neural network (Figure 2J). Analysis of 12 neural signal links showed that the average path length of the links was (Figure 2K). Cross-correlogram between signals from different electrodes was used to characterize the strength of neural network activity, yielding a correlation value of (Figure 2L), indicating synchronization of neural signals across electrodes. Taken together, the results suggest that maturation neural network functions established in Ntoids.
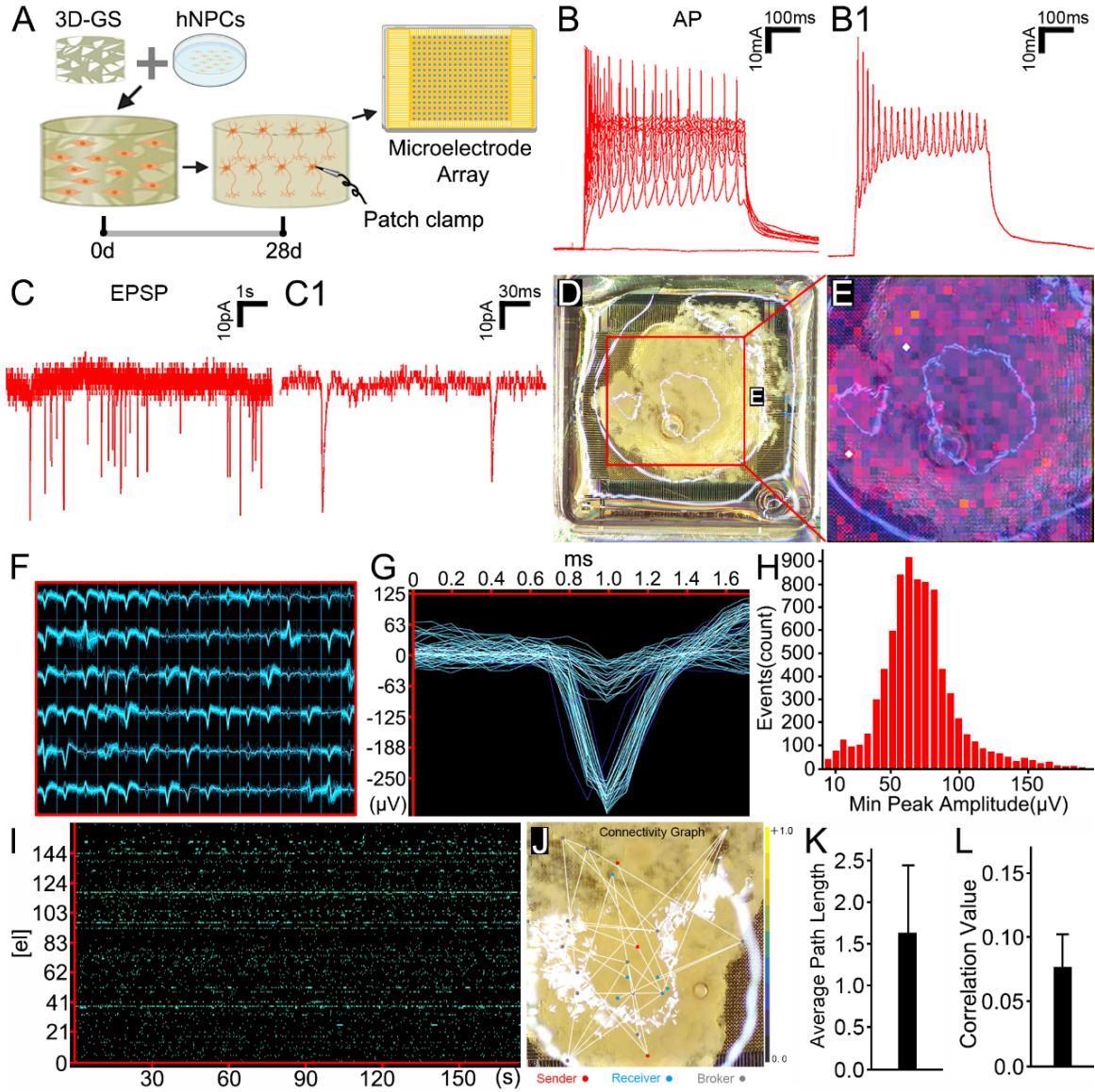
Figure 2. Function maturation and neural network formation of Ntoids. (A) Schematic diagram shows a 28-day cultured Ntoid used for patch clamp and microelectrode array analysis. Whole cell patch-clamp recording evoked action potentials (B-B1) and miniature excitatory postsynaptic currents (EPSCs, C-C1) of neurons after seeding for 28 days. (D) A Ntoid mounted on a high-density microelectrode array. (E) Heat map displays real-time electrical activity on the chip. (F) Summary of all electrodes with spike signals. Each small window represents the spike signal recorded during this period. (G) Typical spike waveform diagram shows the maximum spike amplitude reached . (H) Spike amplitude distribution indicates amplitude level is peaked around . (I) Raster plot reveals clusters of burst firing within Ntoid. (J) Overlay of a neural network connectivity map and a Ntoid. Red dots indicate signal-sender neurons, blue dots signal-receiver neurons, and gray dots signal-broker neurons. Lines represent links between neurons. (K) Average path length of links ( . (L) Cross-correlogram indicates the correlation value for neural network activity.
¶ 3.3. The TK-GCV system effectively eliminated proliferative cells without affecting neuronal tissueoid formation
To eliminate proliferative cells, iPSCs carrying the thymidine kinase gene (hiPSCs-GFP-TK) were constructed by lentiviral gene (GFP-TK-blasticidin) transfection (Figure 3A). After 1–2 rounds of blasticidin selection, purified hiPSCs-GFP-TK expressed green fluorescence (Figure 3B). The pharmacological efficacy of TK was validated by dose-response analysis after adding the non-toxic substrate GCV to the culture medium of either hiPSCs-TK-GFP or hiPSCs-TK-GFP derived neural progenitors (hNPCs-GFP) (Figure 3B-I). Analysis showed that GCV effectively eliminated the majority of hiPSCs-GFP-TK (survival rate ; Figure 3c2, E) or hNPCs-GFP-TK (survival rate ; Figure 3g2, I ); while GCV further reduced the survival of hiPSCs-GFP-TK (survival rate ; Figure 3c3, E) or hNPCs-GFP-TK ; Figure 3g3, I). Notably, was considered to be safe for wild-type hiPSCs or hNPCs, as both of these cells showed a negligible change in their population following drug exposure (survival rate and , respectively; Figure 3d3, E, h3, I). Introduction of the gene did not affect the phenotypic profiles or neural differentiation of hNPCs-TK-GFP, as evidenced by the neural rosette/sphere formation and expression of stem/progenitor markers such as Sox1 , Figure 3J, M), Pax6 , Figure 3K, M) and hNestin , Figure 3L, M) in a fashion similar to wild-type hNPCs (Figure 3M). Similarity, did not affect the neuronal differentiation of hNPCs-TK-GFP, as mature neuronal markers such as the proportions of NeuN , Figure 3N, Q), MAP2 , Figure 3O, Q) or NF200 , Figure 3P, Q) was similar when compared between neurons and neurons (Figure 3Q). Notably, although the stepwise Ntoid construction protocol yielded an extremely low residual iPSCs population , Supplementary Figure 3R), the administration of GCV eliminated almost half of the residual pluripotent cells (Figure 3R) with no discernible effect on the viability of terminal differentiated neurons (Supplementary Figure 2S).
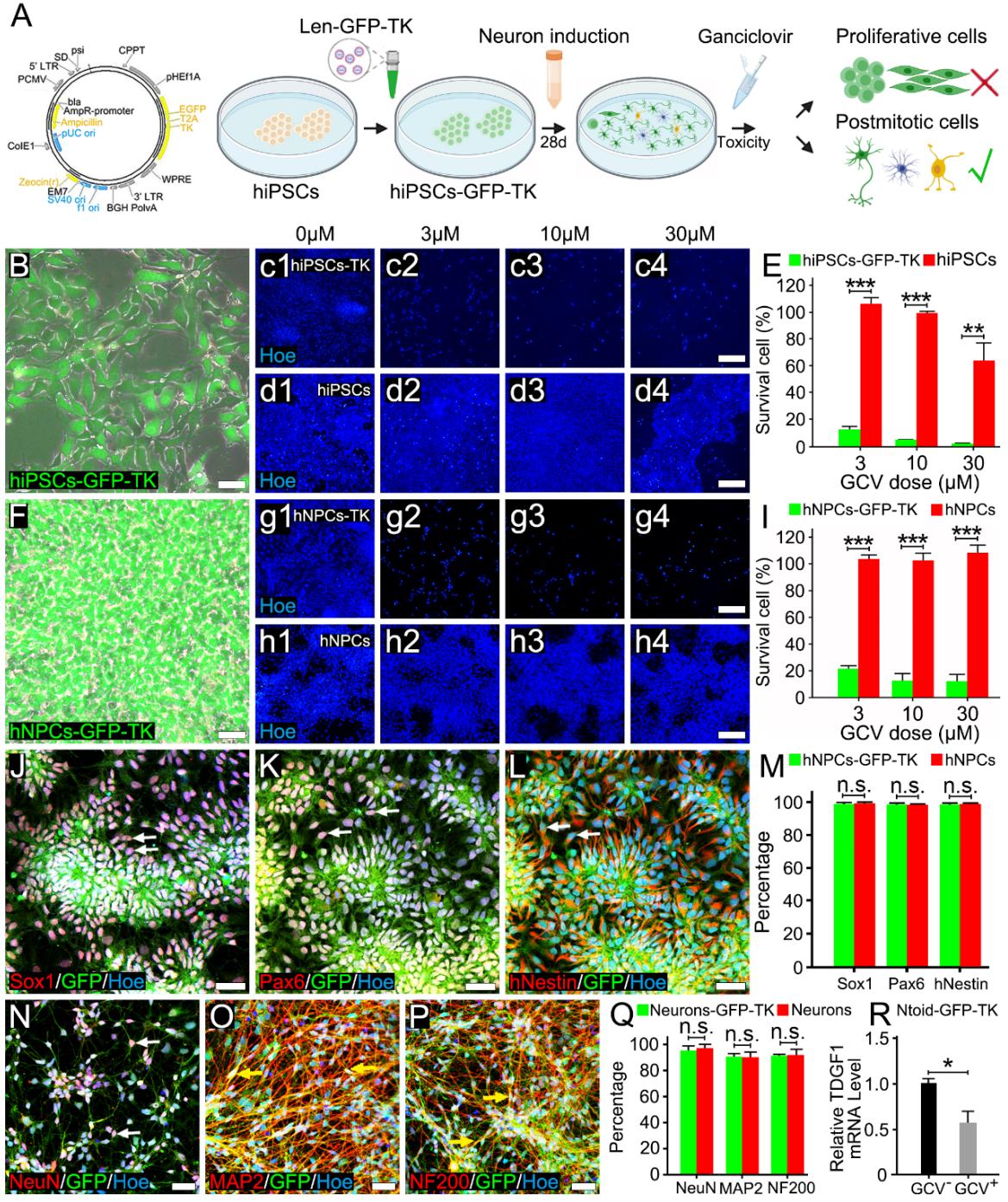
Figure 3. The TK-GCV system eliminated proliferative cells without affecting hiPSC-derived neuron formation. (A): Schematic diagrams showing the construction of hiPSCs-GFP-TK and the elimination of proliferative cells. (B): Morphology of hiPSCs-GFP-TK clones in culture under inverted phase contrast microscopy. (c1-d4): Immunofluorescence staining showing that the TK-GCV system effectively eliminated hiPSCs-GFP-TK after adding the substrate GCV compared with hiPSCs. (E): Bar chart showing the statistically significant difference in survival rate for each group $\stackrel { * * * } { p } < 0 . 0 0 1 $ ). Each experiment was repeated three times with five biological replicates per repeat; for the , for the and for the . (F): Morphology of hNPCs-GFP-TK in culture under inverted phase contrast microscopy. (g1-h4): Immunofluorescence staining showing that the TK-GCV system effectively eliminated hNPCs-GFP-TK after adding the substrate GCV compared with hNPCs. (I): Bar chart showing the statistically significant difference in survival rate for each group ). Each experiment was repeated three times with five biological replicates per repeat; for the , 0.0001 for the and for the . (J-L): Immunofluorescence staining showing the high expression of Sox1 (arrows, J), Pax6 (arrows, K) and hNestin (arrows, L) in hNPCs-GFP-TK. (M): Bar chart showing there was no significant difference in the proportion of , Pax6+ and hNestin+ cells in cells between hNPCs-GFP-TK and hNPCs. Each experiment was repeated three times with five biological replicates per repeat; for the Sox1, for the pax6 and for the hNestin. (N-P): Immunofluorescence staining showing the high expression of NeuN (arrows, N), MAP2 (arrows, O) and NF200 (arrows, P) in the neurons-GFP-TK. (Q): Bar chart showing that there was no significant difference in the proportion of NeuN , and cells in cells between neurons-GFP-TK and neurons. Each experiment was repeated three times with five biological replicates per repeat; 0.074 for the NeuN, for the MAP2 and for the NF200. ®: Bar chart showing that GCV eliminated half of the residual relative mRNA level. Each experiment was repeated three times; . Scale bars (B, F, J-L and N-P), (c1-d4 and g1-h4).
¶ 3.4. Transplantation of hiPSC-derived neuronal tissueoid improved hindlimb motor function
Motor function was assessed by open field and inclined grid motor testing, and electrophysiology tests in rats following Ntoid transplantation (Ntoid group), hNPC-seeded 3D-GS transplantation (hNPCs group) or no cell-seeded 3D-GS transplantation (GS group). Rats received transected SCI in the T10 segment resulting in BBB scores of 0, thus indicating the complete loss of hindlimb motor function. Gradual recovery of hindlimb motor function was observed in all rats. In particular, rats in the NToid group presented significant motor function recovery with a BBB score of at eight weeks after SCI (Figure 4B1-B3, H), thus indicating the recovery of extensive movement up to two joints of the hind limbs. In contrast, the recovery of motor function was mild in the hNPCs group (BBB score: , Figure 4D1-D3, H); in the GS group the BBB score was (Figure 4F1-F3, H) eight weeks after SCI (Figure 4F, H), thus suggesting that the transplantation of hNPCs by GS-based delivery did not provide extra benefit to motor functional recovery when compared with the GS group. The inclined grid test further validated the recovery of voluntary movement in manner other than reflex. Indeed, frequent plantar placements with occasional inter-hindlimb coordination was observed in the Ntoid group (Figure 4C1-C3), in contrast to passive movement without plantar placement of the hindlimbs in the hNPCs group (Figure 4E1-E3) or GS groups (Figure 4G1-G3) at eight weeks after SCI. Recovery of neuronal connectivity was evaluated by the detection of cortical motor-evoked potentials (CMEPs) obtained from transcranial electrophysiological recordings. Analysis showed that the latency of CMEPs was significantly shorter in the NToid group ) compared with the hNPCs group and the GS group ; Figure 4I, J), thus suggesting faster electrical transmission spreading across the injury/graft site of the spinal cord. In addition, the amplitude and the area of CMEPs were significantly larger in the NToid group ( , , respectively) compared with the hNPCs group , , respectively) and the GS group ( , , respectively; Figure 4I, K-L), thus suggesting that a larger number of neurons in the motor neural pathway were electrically activated. Although the transplantation of hNPCs by GS-based delivery did not provide extra benefit to motor function recovery when compared with the GS group (Figure 4H), the amplitude and the area of CMEPs were significantly larger in the hNPCs group when compared with the GS group (Figure 4K-L), thus suggesting that more neurons in the motor neural pathway were electrically activated. Collectively, the therapeutic efficacy of Ntoids was superior than transplanted hNPCs or the GS scaffold with regards to motor function recovery of the hindlimbs within eight weeks.
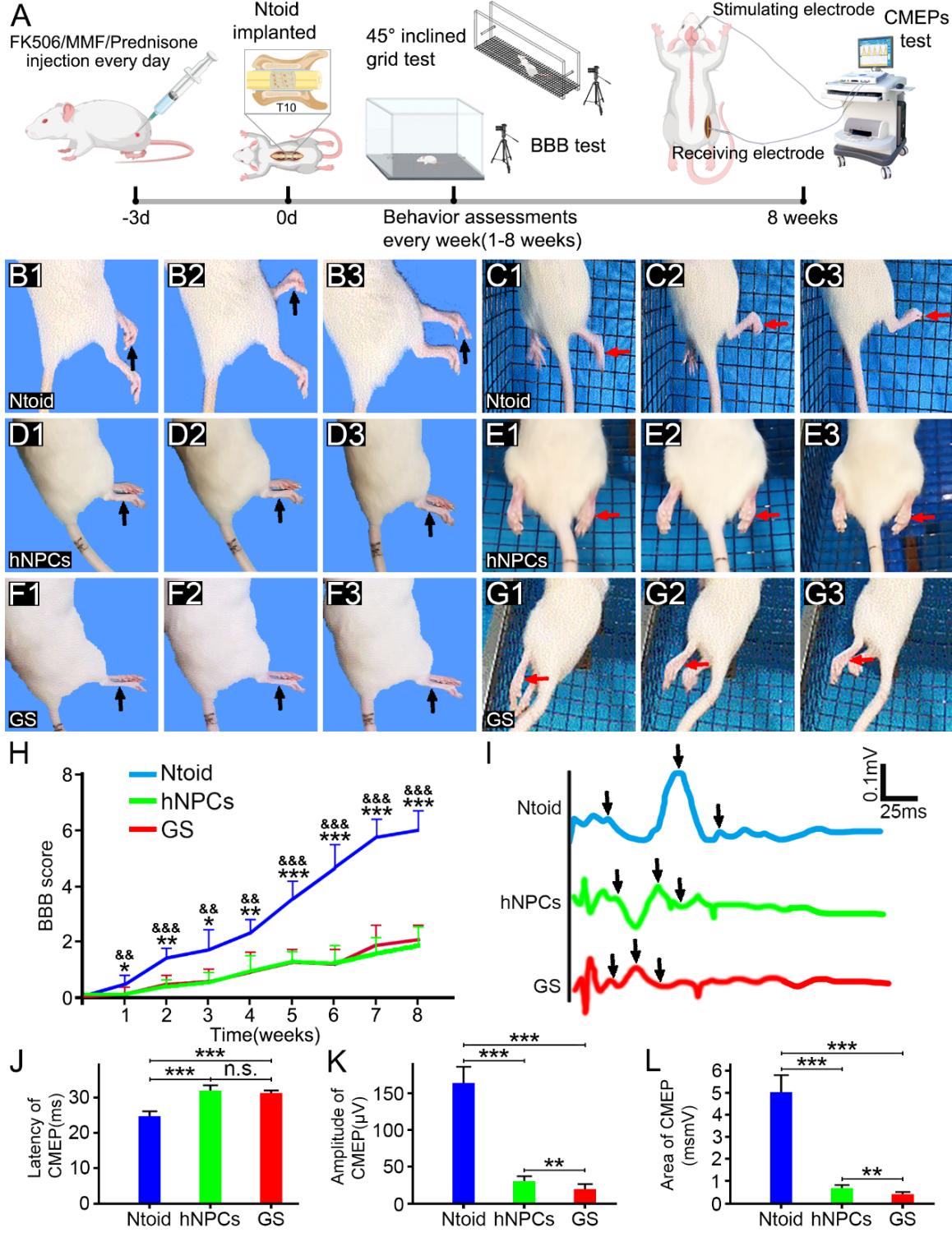
Figure 4. Analyses of behavior and electrophysiological outcome. (A): Schematic diagrams of the surgical and transplantation processes, BBB test, inclined grid test and electrophysiological records for the three groups of rats. (B1-B3, D1-D3 and F1-F3): Open-field observation in the Ntoid (B1-B3), hNPCs (D1-D3) and GS (F1-F3) groups of rats. (H): BBB scoring significantly increased for hindlimb movement functionality following Ntoid ) transplantation and indicate statistical significance when compared with the hNPCs group , , 0.000193, 0.005, 0.001, , , , 0.0001 at 1w, 2w, 3w, 4w, 5w, 6w, 7w and 8w respectively) and , and 0.001 indicate statistical significance when compared with the GS group , , 0.003, 0.01, 0.004, 0.000202, , , at 1w, 2w, 3w, 4w, 5w, 6w, 7w and 8w respectively)).(C1-C3, E1-E3 and G1-G3): Representative images of the hindlimbs (arrows) of a rat climbing the inclined grid in the Ntoid (C1-C3), hNPCs (E1-E3) and GS (G1-G3) groups. (I): Potential diagrams of the electrophysiological assessment of the hindlimbs in each group. (J-L): Bar charts showing a comparison of average latency( for Ntoid vs hNPCs and GS group and for hNPCs vs GS group), amplitude for Ntoid vs hNPCs and GS group and for hNPCs vs GS group) and the area of for Ntoid vs hNPCs and GS group and for hNPCs vs GS group). There was significant difference between Ntoid ) outcomes and those of the or groups and .
¶ 3.5. Robust donor survival in Ntoid group
Eight weeks after transplantation, many cells with green fluorescence were observed within the injury/graft site of spinal cords, indicating the remarkable survival of donor cells carrying TK-GFP. Indeed, the cells suffused the entire injury/graft site and did not present discernible long-distance cell migration to the areas rostral and caudal to the injury/graft site (Figure 5A). Immunostaining of human nuclei further validated that these cells were human-origin (Figure 5D). As with the distribution of cells, hNu immunopositive cells were also located in the injury/graft site (Figure 5D, E-F). Stereological analysis showed that of cells within the injury/graft site were cells, thus suggesting that the surviving donor cells constituted the majority of the cell population in this area (Figure 6J). On the other hand, donor cells in the hNPCs group were scattered and formed focal clusters at the injury/graft site (Figure 5B-b). The density of cells within the injury/graft site in the Ntoid group was significantly larger than that in the hNPCs group vs. , Figure 5C), suggesting that Ntoid transplantation yielded more donor cell survival relative to hNPCs transplantation. Notably, while the transplantation of hNPCs seeded in the GS scaffold yielded teratoma formation in one rat in the hNPCs group (1/10), no teratoma formation was encountered in the Ntoid group (Supplementary Figure 3).
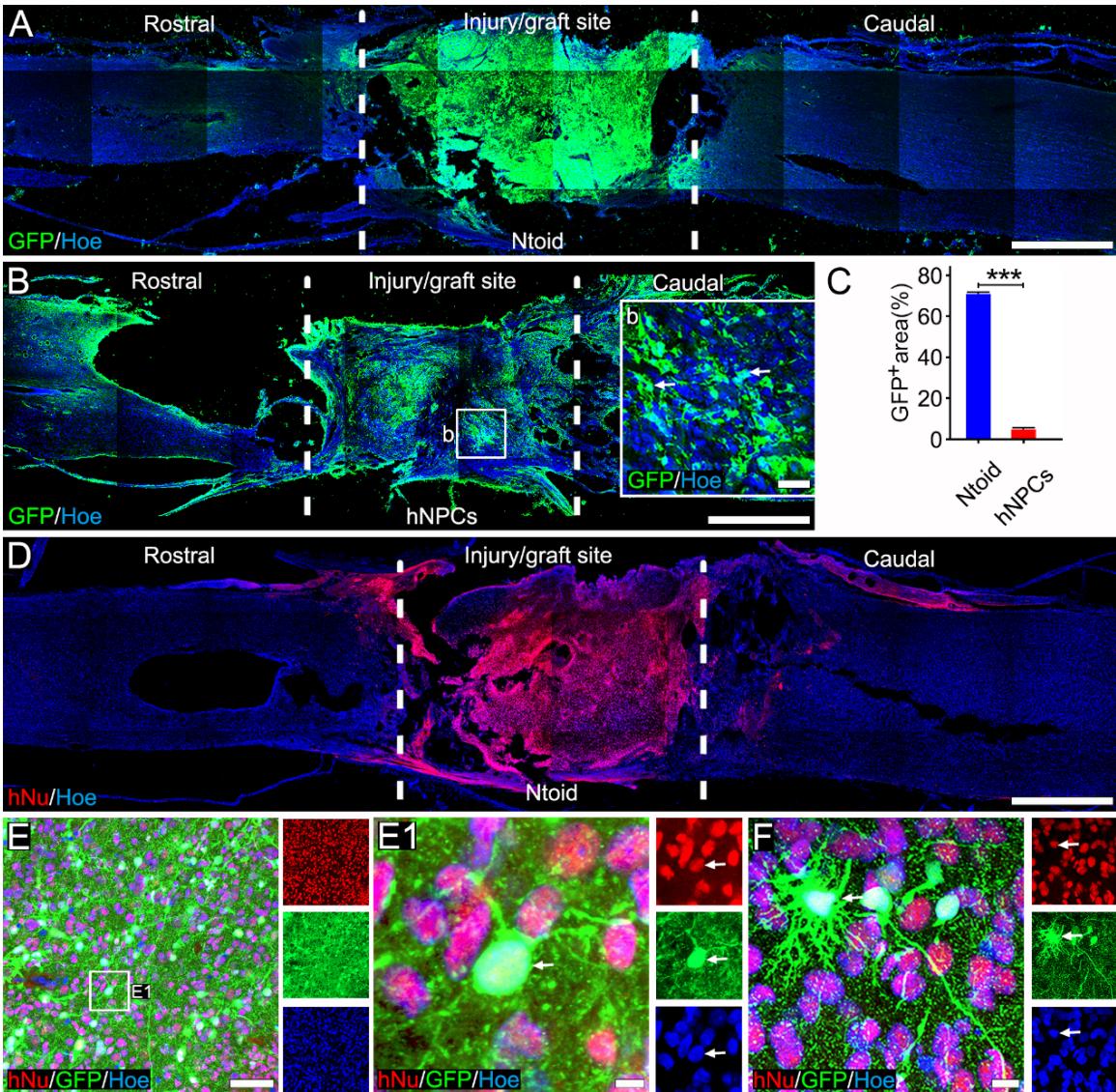
Figure 5. Survival of transplanted hiPSC-derived cells at the injury/graft site. (A-B): Immunofluorescence staining showing the survival of cells at the injury/graft site in the Ntoid (A) and hNPCs (B) groups. (b): A higher magnification image showing cells (arrows) at the injury/graft site in the hNPCs group. ©: Bar chart showing the area in the injury/graft site in the Ntoid and hNPCs groups , five animals/group, mean of 3-5 sections/animal; ). (D): Immunofluorescence staining showing the survival of cells at the injury/graft site in the Ntoid group. (E-F): A representative image showing cells co-expressing hNu and GFP (E-F) at the injury/graft site in the Ntoid group; higher magnification images (E1) of the boxed area shown in (E). Scale bars (A, B and D), (b and E), and (E1 and F).
¶ 3.6. Intensive innervation of the injury/graft site endowed by neuronal tissueoids
Eight weeks after neuronal tissueoid transplantation, the injury/graft site in the spinal cord was remarkedly re-innervated, as evidenced by intense immunosignals of Tuj1 and neurofilament (NF), both of which are common markers for nerve fibers. The fluorescent intensity of NF in the injury/graft site was in the Ntoid group (Figure 6A-A1, G); this was significantly higher than that in the hNPCs group , Figure 6B-B1, G) or the GS group , Figure 6C-C1, G). Similarly, the fluorescent intensity of Tuj1 at the injury/graft site was also higher in the Ntoid group , Figure 6D-D1, H) relative to that in the hNPCs group , Figure 6E-E1, H) or the GS group ( , Figure 6F-F1, H). In addition, immunostaining of NeuN, a marker of mature neurons, revealed that a large population of hiPSCs-derived neurons survived at the injury/graft site in the Ntoid group (Figure 6I-I1). Indeed, of cells at the injury/graft site were NeuN+. Analysis revealed that of all cells expressed NeuN (Figure 6J), thus suggesting that the majority of donor cells represented a neuronal population similar to their composition prior to transplantation. Furthermore, the population of and hNu- cells was scarce at the injury/graft site, thus suggesting that the dominant neurons in this area were donors. Phenotypic characterization of these neurons showed that most of the donor cells were (Figure 6K-k1), and that cells (Supplementary Figure 4A-a2) or cells (Supplementary Figure 4B-b2) were scarce. This may suggest that donor cells maintained cholinergic rather than glutamatergic or GABAergic differentiation at the injury/graft site.
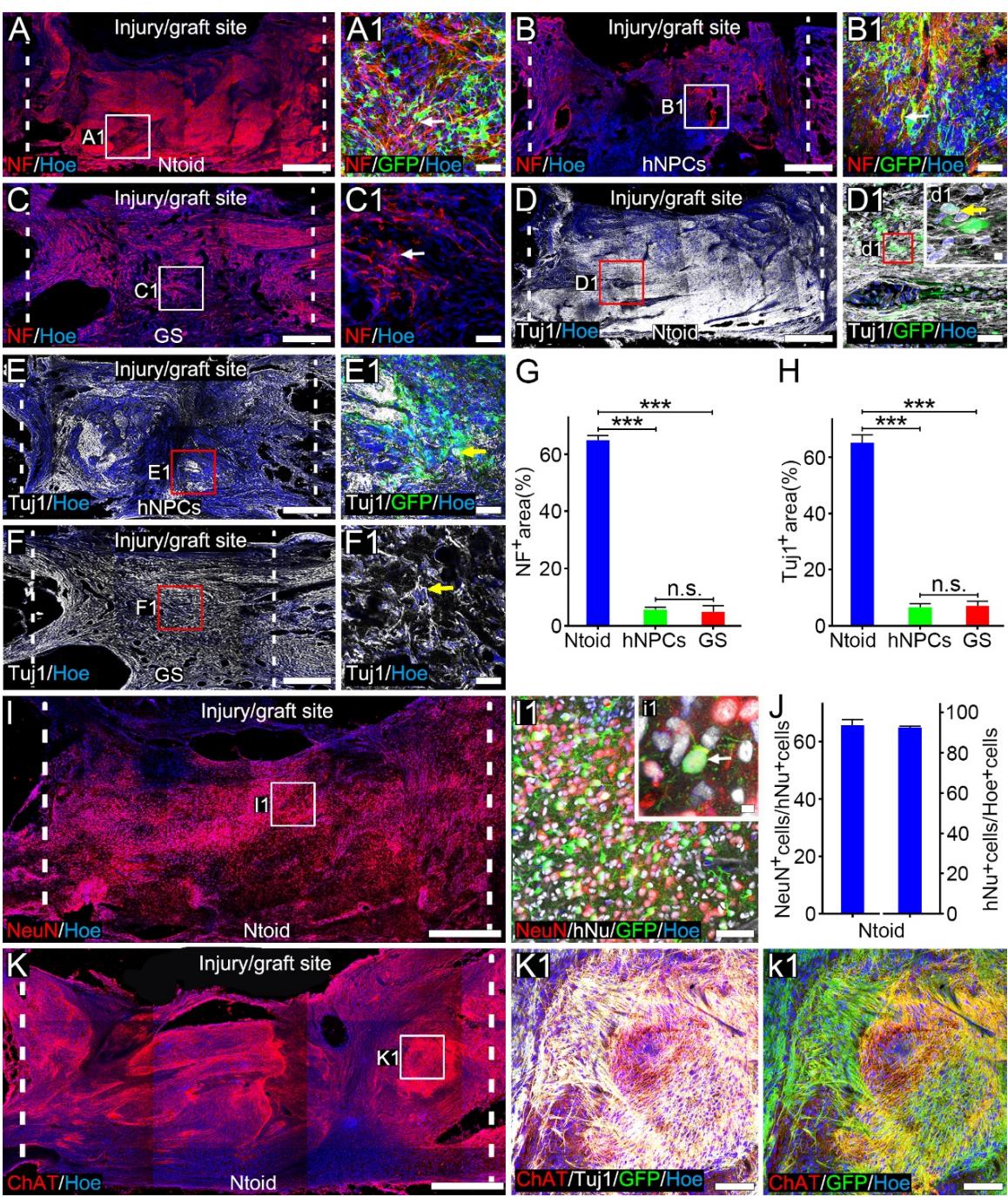
Figure 6. Phenotypic characterization of hiPSC-derived neurons at the injury/graft site. (A-C): Immunofluorescence staining showing NF-positive cells at the injury/graft site in the Ntoid (A), hNPCs (B) and GS © groups of rats. (A1-C1): Representative image showing cells (arrow) at the injury/graft site in the Ntoid (A1), hNPCs (B1) and GS (C1) groups. (D-F): Immunofluorescence staining showing cells at the injury/graft site in the Ntoid (D), hNPCs (E) and GS (F) groups. (D1-F1): Representative image showing cells (arrows) at the injury/graft site in the Ntoid (D1), hNPCs (E1) and GS (F1) groups. Insert (d1): a higher magnification image of the boxed area, demonstrating a T cell (arrow). (G): Bar chart showing the area at the injury/graft site in three groups , five animals/group, mean of 3-5 sections/animal; for Ntoid vs hNPCs and GS group and for hNPCs vs GS group). (H): Bar chart showing the area at the injury/graft site in three groups 0.001, five animals/group, mean of 3-5 sections/animal; for Ntoid vs hNPCs and GS group and for hNPCs vs GS group)). (I): Immunofluorescence staining showing NeuN-positive cells at the injury/graft site in the Ntoid group. (I1): Representative image showing cells co-expressing NeuN and hNu or GFP at the injury/graft site. Insert (i1): higher magnification of the boxed area in (I), demonstrating a neuron (arrow). (J): Bar chart showing the ratio of cells in Hoe+ cells and the ratio of NeuN+ cells in cells at the injury/graft site in the Ntoid group. (K): Immunofluorescence staining showing ChAT+ cells at the injury/graft site in the Ntoid group. (K1-k1): Representative image showing the cells co-expressing ChAT, Tuj1 and GFP (arrows) at the injury/graft site in the Ntoid group. Scale bars (A-F, I and K), (A1-F1 and I1), (K1 and k1) and (d1 and i1).
¶ 3.7. Astrocytic differentiation in transplanted Ntoids
Considering the persistent gliosis after SCI, we performed immunofluorescence staining of glial fibrillary acidic protein (GFAP) to assess the distribution of astrocytes after SCI. The distribution of reactive astrocytes at the interface between the injury/graft site and the residual spinal cord tissue was thought to be involved in the formation of glial scarring during chronic SCI, as we observed in the GS group. Few GFAP+ astrocytes were observed at the injury/graft site (Supplementary Figure 5). In contrast, many GFAP-positive astrocytes were observed at the injury/graft site in the Ntoid group (Figure 7A). GFAP+/hNu+ cells were observed at the injury/graft site (Figure 7B), thus suggesting that a portion of the surviving donor cells differentiated into astrocytes. In addition, many neurons were distributed in the GFAP+ area (Figure 7C); in addition, nerve fibers grew into and penetrate the area (Figure 7D-D1), thus suggesting that astrocytes at the injury/graft site may act as a “bridge” rather than a “barrier” to regenerating axons. In addition, we observed feet-like structures on hiPSCs-derived astrocytes at the injury/graft site; these made close contacts with vascular endothelial cells (Figure 7E-e2). This suggested that donor-derived astrocytes may participate in the formation of blood-spinal cord-barrier. On the other hand, astrocytic differentiation of donor cells was also detected in the hNPCs group, and hNPCs tended to be more differentiated into astrocytes other than neurons (Figure 7F-H).
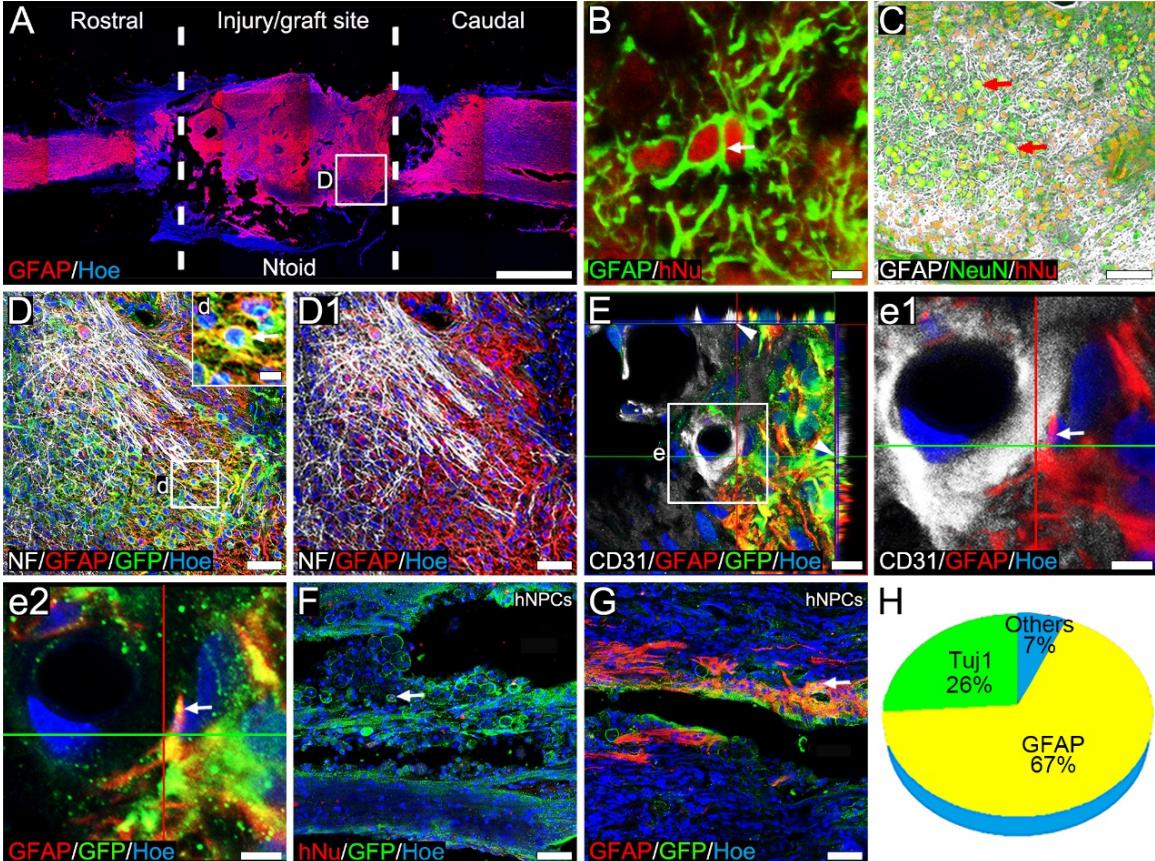
Figure 7. The transplantation of Ntoids regulated the functionality of astrocytes at the injury/graft site. (A): Immunofluorescence staining showing the distribution of astrocytes at the injury/graft site of the spinal cord in the Ntoid group. (B): High magnification image showing the co-expression of hNu and GFAP at the injury/graft site. ©: Immunofluorescence staining showing the morphology and distribution of astrocytes and neurons at the injury/graft site in the Ntoid group. (D-D1): Immunofluorescence staining showing that the regenerated nerve fibers were distributed in the GFAP+ region of the injury/graft site. (E-e2): Immunofluorescence staining showing that the hiPSCs-derived astrocyte formed feet-like structures making contact with vascular endothelial cells at the injury/graft site. (E-G): Image showing cells co-expressing GFP and hNu (E) or GFAP(F) at the injury/graft site in the hNPCs group. (H): Pie chart showing the ratio of or cells of cells at the injury/graft site in the hNPCs group. Scale bars ; (B, d and e1-e2); (C, D-D1, F, and G) and .
The integration of donor neurons with host neural pathways was evaluated by detecting the connection between donor neurons and host descending/ascending nerve fibers that had regenerated to the grafts, and between the donor neuronal nerve fibers/dendrites and host residual neurons (Figure 8A). The transplanted neurons in Ntoids extended long processes towards the area caudal to the injury/graft site of the spinal cord; there were more caudal processes than rostral fibers. Co-labeling with NF GAP43 markers in most processes suggested that these were long nerve fibers from donor neurons (Figure 8B), with sustained capability for significant growth and elongation (Figure 8E). The density of donor nerve fibers, as labeled by GFP, decreased along with increasing distance. There were donor nerve fibers at 1mm caudal to the injury/graft site; this higher than the number of fibers at ( ) or at ) (Figure 8B-D). A small number of donor nerve fibers projected into the L1-L2 lumbar spinal cord, making close contacts with dorsal horn neurons (Supplementary Figure 6). Closer investigation showed that donor nerve fiber terminals expressing Syn, formed bouton-like profiles on the soma surface of ChAT+ neurons in the lumbar spinal cord (Figure 8F-f2), indicating the possibility of synapse formation. Supraspinal nerve fiber regeneration and synaptic integration with grafted neurons are crucial for repairing disrupted brain-spinal cord connections. nerve fibers were used as a representative of brain-derived descending nerve fibers to investigate connectivity with donor neurons in Ntoids. Although limited in number, nerve fibers regenerated into the injury/graft site and formed connections with grafted cells. The bouton-like structure expressed Syn, thus suggesting that nerve fibers may establish synaptic connections with grafted neurons (Figure 8G-g2). Furthermore, a subset of nerve fibers made contacts with donor cells located at the center of the injury/graft site (Figure 8H-H1). The close contacts between CGRP+ nerve fibers and donor cells expressing Syn may indicate synapse formation (Figure 8H1). Some donor nerve fibers extending to the area caudal to the injury/graft site made close contacts with nerve fibers (Figure 8I-I1). Collectively, these results suggest that survival donor neurons could integrate into host neural pathways to repair the disrupted neural pathways after SCI.

Figure 8. Integration of a transplanted Ntoid into host neural pathways in injured spinal cord. (A): Schematic diagrams of the integration of donor neurons with host neural pathways. (B-C): Immunofluorescence staining showing long processes (arrow) extending from donor neurons were observed in the area caudal to the injury/graft site. Insert (b): a higher magnification image of the boxed area in (B), demonstrating or nerve fibers (arrow) at (B) and © distances from the injury/graft site. (D): Bar chart showing the density of nerve fibers in the area caudal to the injury/graft site. (E): Immunofluorescence staining showing 29 the co-localization of long nerve fibers with growth cones in the area caudal to the injury/graft site. (F): A representative image showing host ChAT+ neurons, presynaptic terminals and donor nerve fibers in the area caudal to the injury/graft site. (F1-f2): Higher magnification images of the boxed area in (F), demonstrating an abundance of presynaptic terminals (arrow) and a small number of nerve fiber terminals (arrows) distributed and making contact on/with the soma surface of ChAT+ neurons. (G): Immunofluorescence staining showing regenerated nerve fibers, presynaptic terminals, and neurons at the injury/graft site. (G1-g2): Higher magnification image of the boxed area in (G), demonstrating the nerve fiber and presynaptic terminals contacting nerve fibers (arrow). (H): Immunofluorescence staining showing regenerated CGRP+ nerve fibers, presynaptic terminals, and neurons and their nerve fibers at the injury/graft site. (H1): Higher magnification image of the boxed area in , demonstrating the nerve fiber and presynaptic terminals contacting dendrite/nerve fibers (arrow). (I): Immunofluorescence staining showing a nerve fiber extending to the area caudal to the injury/graft site. (I1): Higher magnification image of the boxed area in (H), demonstrating a nerve fiber making contact with a nerve fiber (arrow). Scale bars (B, C, E, H and I), (G), (b and F) and (F1-f2, G1-g2, H1 and I1).
¶ 4. Discussion
Encouraged by the successful construction of human iPSC-derived neuronal tissueoids in vitro, we investigated the efficacy of transplanted Ntoids for SCI repair in a rat model of complete SCI. First, functional synaptic connections, a high neuronal differentiation rate and an extremely low residual pluripotent cell population were validated in Ntoids in vitro. This stepwise tissueoid construction process, together with the TK-GCV suicide gene-based safe-cell system, eliminated the uncertainty of hiPSCs differentiation and improved the safety of hiPSCs for transplantation. Second, a multi-modal assessment battery including the BBB test and cortical motor-evoked potentials (CMEPs) revealed the continuous improvement of movement in the paralyzed hindlimbs of rats following Ntoid transplantation. Third, histological analysis revealed that hiPSC-derived neurons survived for up to eight weeks and formed synaptic connections with host neurons via long nerve fibers/dendrites. The host regenerating nerve fibers grew into the injury/graft site and formed synapses with grafted neurons, thus suggesting that the surviving hiPSC-derived neurons could integrate into host neural pathways. Therefore, the donor neurons in Ntoids served as a “neuronal relay” to restore neural pathways[34], thus promoting the recovery of motor function in the paralyzed hindlimbs of rats with complete SCI.
Patient safety after transplantation is a key priority for iPSC-based cell therapy. Recent progress in chemical reprogramming[35] has effectively tackled a number of safety issues related to iPSCs, including tumorigenicity caused by reprogramming factors[36,37], or genetic mutation[38,39], thus enabling the successful clinical application of iPSC-derived therapeutic cells[40]. However, uncertain post-transplantation differentiation arising from the nature of pluripotency remains a key hurdle for iPSC-based specific tissue repair. Of these, teratoma formation is considered the most undesirable donor fate, and cannot be addressed simply by increasing quality control at the pluripotent stage. The selective removal of undifferentiated cells by the HSV-TK system[41] or by highly purified iPSCs-derived neural progenitor cells (NPCs )[42], have been proven to effectively improve the safety of iPSCs transplantation. Therefore, in the current study, we developed a combined protocol to engage stepwise neuronal tissueoid construction to acquire a terminal-differentiated population and designed a TK-GCV system to purify the post-mitotic population. Our analysis showed that more than of hiPSCs were induced into hNPCs in vitro. These cells continuously matured and achieved up to terminal differentiation towards neurons in a 3D GS scaffold over 14 to 28 days, yielding an extremely low pluripotent population ). The application of GCV provided extra efficacy in eliminating proliferating cells from among neurons in tissueoids without affecting neuronal viability. Therefore, we successfully established a dual-safeguard mechanism to ensure the safety of iPSC-derived therapeutic cells for SCI repair, which may also provide an effective strategy for other iPSC-based applications.
Our research provided novel methodology for human mature neuron transplantation to repair an injured nervous organ. Indeed, NPCs seeded in a 3D biocompatible scaffold for neuronal maturation and tissue formation eventually yielded an implantable Ntoid that could repair defects in nervous tissue. It is noteworthy that tissueoid transplantation, in contrast to the delivery of cells seeded in a vehicle bioscaffold, may offer a novel cell therapy regimen. Indeed, the neuronal tissueoid culture process aligned with the “four-dimensional (4D) biology” principle[43], which involves a time period in 3D culture in the context of executing a specific function, which could subsequently encourage dynamic interactions between cells and the extracellular matrix (ECM) in a culture environment, leading to the formation of a homeostatic tissue-like entity with specific repair function. Indeed, the results from our current study showed that the transplantation of Ntoids conferred significantly better donor survival and neuronal differentiation and no presence of teratoma formation, relative to 3D-GS based NPC transplantation. The underlying mechanisms contributing to these superior therapeutic outcomes are four-fold. First, hNPCs continuously matured in a 3D scaffold to achieve significant terminal differentiation towards neurons, thus guaranteeing a desirable hiPSC fate for neural repair. Second, specific neuronal phenotypes could be induced prior to transplantation[44,45], offering the possibility of “precision treatment” for spinal cord injury by using targeted neuronal types. In the current study, excitatory neurons were used to repair disconnected neural pathways following SCI; this is because intraspinal excitatory inputs are essential for motor function[46]. In the future, function-related neuronal subtypes[47,48] should be transplanted and investigated for their ability to induce better functional recovery. Third, the homeostatic niche formed by the dynamic interactions between the cells and ECM over time provided spatiotemporal cues to reshape cell behavior[49]. This may facilitate the survival of donor cells in the harsh post-SCI milieu and integrate with host neural pathways. Fourth, the 2 to 4 weeks in vitro construction time for Ntoids provides a window for the real-time monitoring of batch quality; this is a useful avenue for quality control and off-the-shelf cell production[50]. Collectively, these findings indicate that the construction of Ntoids could be an effective method with which to transplant human mature neurons. The temporal window during tissue morphogenesis can be effectively leveraged to enable the construction of functionally specialized therapeutic cell subpopulations via diverse developmental induction strategies, and the implementation of randomized cell quality sampling protocols.
The recovery of voluntary motor function is one of the fundamental goals of SCI treatment. In the present study, the recovery of voluntary motor function in multiple joints, including the hip, knee and ankle was first presented just four weeks after NToid transplantation with sustained and incremental motor recovery until the end of the study. In contrast, rats receiving NPC grafts did not exhibit meaningful motor function recovery. The presumed reason for this significant change is the substantial survival of transplanted cells in the Ntoid grafts, whereas NPC transplantation led to a greater loss of the transplanted population, as evidenced by GFP reporter-based tracking of donor numbers. Indeed, as executors of repair functions, the quantity of surviving transplanted cells may correlate with neural functional recovery[51]. Therefore, it is safe to conclude that Ntoid delivery effectively protected the transplantation of cells in the hostile microenvironment of the injury site. In addition, the transplantation of NSCs or NPCs preferentially differentiated into astrocytes other than neurons[52] and therefore a combination of multiple neurotrophic factors to promote the survival of donor cells and neuronal differentiation was usually applied in this regimen. However, we observed that NPC/NSC-derived donor cells migrated into the adjacent tissue at a mean rate of month and was accompanied by cell division[53], rendering the risk of ectopic neurogenesis to disturb local neural circuits. On the other hand, the transplantation of neurons was observed only at the injury/graft site, thus ruling out the possibility of ectopic migration. It should be noted that immunomodulatory iPSC-derived neuron transplantation protocols have been developed by overexpressing immunomodulatory transgenes to enable transplant immune evasion[54]. In this study, we adopted a post-transplantation immunosuppression regimen that was aligned with organ transplantation in the clinic, which is essential for xenogeneic neuron transplantation. The use of immunosuppressants further highlighted the hypothesis that Ntoids promoted the survival of transplanted cells; this is because cell loss after NPC transplantation may not stem from excessive immune rejection but rather from the lack of tissueoid niche support for donor cells. Of course, this conclusion requires further supportive evidence. Furthermore, observable motor function improvements at four weeks post-transplantation further highlight the therapeutic advantage of transplanted neurons over NPCs. Previous studies indicated that functional recovery after NPC transplantation occurred at significantly delayed timepoints (e.g., up to 18 weeks)[26], sometimes requiring over one year to manifest motor function improvements[27]. Phenotypic and functional matured donor neurons were inferred to be responsible for motor function recovery. Taken together, our data demonstrate that when compared to NPC transplantation, Ntoid transplantation resulted in greater survival of donor neurons and better motor improvement, thus suggesting superior clinical application prospects for this therapy.
The integration of donor neurons into host neural tissue is fundamental if we are to repair the disrupted neural pathways following SCI. This process requires the formation of synapses between the host and donor neurons. The extension of nerve fibers/dendrites from donor cells is therefore an important anatomical basis for synaptogenesis with host neurons. The post-injury microenvironment is believed to be inhibitory to nerve fiber regeneration. However, recent studies suggested that traditional inhibitory molecules against nerve fibers associated with myelin, such as Nogo[55], myelin-associated glycoprotein (MAG)[56] and oligodendrocyte myelin glycoprotein (OMgp)[57], actually promote the growth of nerve fibers emerging from grafted hiPSCs-derived NPCs via the interaction with cell adhesion molecule neuronal growth regulator 1 (Negr1)[58]. Similarity, long donor nerve fibers extending to the area caudal to the injury/graft site of spinal cord were observed in the present study. These long nerve fibers formed synaptic contacts with host neurons or nerve fibers to promote the reconstruction of neural pathways and circuits. On the other hand, the regenerative transcriptome of motor neurons in the corticospinal tract (CST) regress to an embryonic transcriptional growth state[59] and enable the robust regeneration of corticospinal nerve fibers[5] following SCI in mammalian adults. The embryonic transcriptional growth state can be sustained by grafting embryonic spinal cord-derived neural progenitor cells, leading to the robust regeneration of corticospinal nerve fibers into the grafts[60]. In this study, host supero-spinal nerve fibers regenerated into the graft and formed synaptic connections with donor neurons, thus suggesting that iPSC-derived neuronal tissueoids may help to sustain the embryonic transcriptional growth state in the brain following SCI, in a manner similar to the embryonic spinal cord-derived neural progenitor cells. Indeed, the amicable embryogenesis-like microenvironment formed in the grafted Ntoid permitted the host nerve fibers to regenerate into the injury/graft site of the spinal cord. However, the exact mechanism involved warrants further investigation.
¶ 5. Conclusions
Functional Ntoids derived from hiPSCs were successfully constructed in a 3D culture system. These Ntoids served as “neuronal relay” to restore neural circuitry and promote the recovery of motor function in the paralyzed hindlimbs of rats with SCI. No adverse reactions were observed following transplantation, such as tumorigenesis, an excessive immune response caused by the xenograft, ectopic migration or hyperalgesia. These findings support the safety and efficacy of the transplantation of mature neuronal tissue with safety-regulatory mechanisms to treat SCI and provide a framework for future clinical application using tissue-engineered Ntoids.
¶ CRediT authorship contribution statement
Xiang Zeng, Yuan-shan Zeng, Qing-shuai Wei and Yuan-huan Ma conceptualized the project design. Qing-shuai Wei and Xiang Zeng performed cell culture, animal experiments, microscopic morphology analysis, statistical analyses and generated figures. Yuan-huan Ma, Ke-jun Zhang, Hui-juan Shi, Li-zhi Peng and Jun-hua Wang assisted in the surgery and behavior assessments in the animal experiments. Wen-tao Ma, Qi-xin Hu, Hou-ge Li and Ying Liu performed molecular biology. Ying Ding, Ge Li and Bi-qin Lai conducted formal analysis. Qing-shuai Wei wrote the manuscript with assistance from Xiang Zeng and Yuan-shan Zeng. Xiang Zeng supervised all aspects of the study.
¶ Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
¶ Acknowledgements
This research was supported by grants from the National Natural Science Foundation of China (82371395 and 81891000) to Xiang Zeng, the National Natural Science Foundation of China(82374579) to Ying Ding, Guangdong Natural Science Foundation of Guangdong Province (2024A1515010814) to Xiang Zeng, the Natural Science Foundation of Guangdong Province (2023A1515012211) to Ying Ding, Guangzhou Basic and Applied Basic Research Foundation (2023A03J0626), the Hospital Funding for Developing Stem Cell Reagent and Organoid Platform (GXB202401) and the Startup Grant of Guangdong Provincial Hospital of Chinese Medicine (2022KT1032) to Xiang Zeng; the National Natural Science Foundation of China (81891003) to Yuan-Shan Zeng; the Natural Science Foundation of Henan Province(252300421617) to Qing-Shuai Wei. We thank Professor Bin Jiang (Sun Yat-sen University, Guangzhou, China) for their technical support on the patch-clamp recording and Professor Andy Peng Xiang (Sun Yat-sen University, Guangzhou, China) for providing the hiPSCs in this study.
¶ References
[1] T.M. O’Shea, J.E. Burda, M.V. Sofroniew, Cell biology of spinal cord injury and repair, J Clin Invest 127 (2017) 3259–3270. https://doi.org/10.1172/JCI90608.
[2] R. Ae, T. Dk, H. I, Y. D, Z. X, A. Je, A. Z, K. Sw, W. H, S. Rl, Z. Rd, T. Yd, Defining recovery neurobiology of injured spinal cord by synthetic matrix-assisted hMSC implantation, Proceedings of the National Academy of Sciences of the United States of America 114 (2017). https://doi.org/10.1073/pnas.1616340114.
[3] L. X, Z. Y, C. S, H. S, S. M, C. B, C. X, T. F, W. N, T. Y, W. B, X. Z, Z. S, D. J, Cetuximab modified collagen scaffold directs neurogenesis of injury-activated endogenous neural stem cells for acute spinal cord injury repair, Biomaterials 137 (2017). https://doi.org/10.1016/j.biomaterials.2017.05.027.
[4] B. Fan, Z. Wei, X. Yao, G. Shi, X. Cheng, X. Zhou, H. Zhou, G. Ning, X. Kong, S. Feng, Microenvironment Imbalance of Spinal Cord Injury, Cell Transplant 27 (2018) 853– 866. https://doi.org/10.1177/0963689718755778.
[5] K. Kadoya, P. Lu, K. Nguyen, C. Lee-Kubli, H. Kumamaru, L. Yao, J. Knackert, G. Poplawski, J.N. Dulin, H. Strobl, Y. Takashima, J. Biane, J. Conner, S.-C. Zhang, M.H. Tuszynski, Spinal cord reconstitution with homologous neural grafts enables robust corticospinal regeneration, Nat Med 22 (2016) 479–487. https://doi.org/10.1038/nm.4066.
[6] P. Assinck, G.J. Duncan, B.J. Hilton, J.R. Plemel, W. Tetzlaff, Cell transplantation therapy for spinal cord injury, Nat Neurosci 20 (2017) 637–647. https://doi.org/10.1038/nn.4541.
[7] J.N. Dulin, A.F. Adler, H. Kumamaru, G.H.D. Poplawski, C. Lee-Kubli, H. Strobl, D. Gibbs, K. Kadoya, J.W. Fawcett, P. Lu, M.H. Tuszynski, Injured adult motor and sensory axons regenerate into appropriate organotypic domains of neural progenitor grafts, Nat Commun 9 (2018) 84. https://doi.org/10.1038/s41467-017-02613-x.
[8] M.M.A. de Almeida, K. Goodkey, A. Voronova, Regulation of microglia function by neural stem cells, Front Cell Neurosci 17 (2023) 1130205. https://doi.org/10.3389/fncel.2023.1130205.
[9] S.M. Hosseini, A. Alizadeh, N. Shahsavani, J. Chopek, J.-E. Ahlfors, S. Karimi-Abdolrezaee, Suppressing CSPG/LAR/PTPσ Axis Facilitates Neuronal Replacement and Synaptogenesis by Human Neural Precursor Grafts and Improves Recovery after Spinal Cord Injury, J Neurosci 42 (2022) 3096–3121. https://doi.org/10.1523/JNEUROSCI.2177-21.2022.
[10] R.A. Fricker, M.K. Carpenter, C. Winkler, C. Greco, M.A. Gates, A. Björklund, Site-specific migration and neuronal differentiation of human neural progenitor cells after transplantation in the adult rat brain, J Neurosci 19 (1999) 5990–6005. https://doi.org/10.1523/JNEUROSCI.19-14-05990.1999.
[11] A.C. Lepore, I. Fischer, Lineage-restricted neural precursors survive, migrate, and differentiate following transplantation into the injured adult spinal cord, Exp Neurol 194 (2005) 230–242. https://doi.org/10.1016/j.expneurol.2005.02.020.
[12] Q.-L. Cao, R.M. Howard, J.B. Dennison, S.R. Whittemore, Differentiation of engrafted neuronal-restricted precursor cells is inhibited in the traumatically injured spinal cord, Exp Neurol 177 (2002) 349–359. https://doi.org/10.1006/exnr.2002.7981.
[13] Lei Wang, Jinlu Gan, Zhihao Xu, Tao Huang, Yingchun Zhou, Xiaobing Jiang, Hongyang Zhao, Deqiang Lei, Laminin-Modified Porous GelMA Microspheres Sustain a Pro-Neurogenic Niche for Neural Stem Cells Transplantation in Spinal Cord Injury, Advanced Functional Materials (2025). https://doi.org/10.1002/adfm.202510129.
[14] G. Li, B. Zhang, J.-H. Sun, L.-Y. Shi, M.-Y. Huang, L.-J. Huang, Z.-J. Lin, Q.-Y. Lin, B.-Q. Lai, Y.-H. Ma, B. Jiang, Y. Ding, H.-B. Zhang, M.-X. Li, P. Zhu, Y.-Q. Wang, X. Zeng, Y.-S. Zeng, An NT-3-releasing bioscaffold supports the formation of TrkC-modified neural stem cell-derived neural network tissue with efficacy in repairing spinal cord injury, Bioact Mater 6 (2021) 3766–3781. https://doi.org/10.1016/j.bioactmat.2021.03.036.
[15] B.-Q. Lai, B. Feng, M.-T. Che, L.-J. Wang, S. Cai, M.-Y. Huang, H.-Y. Gu, B. Jiang, E.-A. Ling, M. Li, X. Zeng, Y.-S. Zeng, A Modular Assembly of Spinal Cord-Like Tissue Allows Targeted Tissue Repair in the Transected Spinal Cord, Adv Sci (Weinh) 5 (2018) 1800261. https://doi.org/10.1002/advs.201800261.
[16] X. Zeng, X.-C. Qiu, Y.-H. Ma, J.-J. Duan, Y.-F. Chen, H.-Y. Gu, J.-M. Wang, E.-A. Ling, J.-L. Wu, W. Wu, Y.-S. Zeng, Integration of donor mesenchymal stem cell-derived neuron-like cells into host neural network after rat spinal cord transection, Biomaterials 53 (2015) 184–201. https://doi.org/10.1016/j.biomaterials.2015.02.073.
[17] B.-Q. Lai, J.-M. Wang, J.-J. Duan, Y.-F. Chen, H.-Y. Gu, E.-A. Ling, J.-L. Wu, Y.-S. Zeng, The integration of NSC-derived and host neural networks after rat spinal cord transection, Biomaterials 34 (2013) 2888–2901.
[18] G.-H. Wu, H.-J. Shi, M.-T. Che, M.-Y. Huang, Q.-S. Wei, B. Feng, Y.-H. Ma, L.-J. Wang, B. Jiang, Y.-Q. Wang, I. Han, E.-A. Ling, X. Zeng, Y.-S. Zeng, Recovery of paralyzed limb motor function in canine with complete spinal cord injury following implantation of MSC-derived neural network tissue, Biomaterials 181 (2018) 15–34. https://doi.org/10.1016/j.biomaterials.2018.07.010.
[19] B.-Q. Lai, M.-T. Che, B. Feng, Y.-R. Bai, G. Li, Y.-H. Ma, L.-J. Wang, M.-Y. Huang, Y.-Q. Wang, B. Jiang, Y. Ding, X. Zeng, Y.-S. Zeng, Tissue-Engineered Neural Network Graft Relays Excitatory Signal in the Completely Transected Canine Spinal Cord, Adv Sci (Weinh) 6 (2019) 1901240. https://doi.org/10.1002/advs.201901240.
[20] J. Yu, M.A. Vodyanik, K. Smuga-Otto, J. Antosiewicz-Bourget, J.L. Frane, S. Tian, J. Nie, G.A. Jonsdottir, V. Ruotti, R. Stewart, I.I. Slukvin, J.A. Thomson, Induced pluripotent stem cell lines derived from human somatic cells, Science 318 (2007) 1917–1920. https://doi.org/10.1126/science.1151526.
[21] K. Takahashi, K. Tanabe, M. Ohnuki, M. Narita, T. Ichisaka, K. Tomoda, S. Yamanaka, Induction of pluripotent stem cells from adult human fibroblasts by defined factors, Cell 131 (2007) 861–872. https://doi.org/10.1016/j.cell.2007.11.019.
[22] K. Takahashi, S. Yamanaka, Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors, Cell 126 (2006) 663– 676. https://doi.org/10.1016/j.cell.2006.07.024.
[23] Y. Kobayashi, Y. Okada, G. Itakura, H. Iwai, S. Nishimura, A. Yasuda, S. Nori, K.
Hikishima, T. Konomi, K. Fujiyoshi, O. Tsuji, Y. Toyama, S. Yamanaka, M. Nakamura, H. Okano, Pre-evaluated safe human iPSC-derived neural stem cells promote functional recovery after spinal cord injury in common marmoset without tumorigenicity, PLoS One 7 (2012) e52787. https://doi.org/10.1371/journal.pone.0052787.
[24] O. T, N. N, K. J, T. O, S. M, S. S, K. Y, M. M, N. M, O. H, Treatment with a Gamma-Secretase Inhibitor Promotes Functional Recovery in Human iPSC- Derived Transplants for Chronic Spinal Cord Injury, Stem Cell Reports 11 (2018). https://doi.org/10.1016/j.stemcr.2018.10.022.
[25] P. Lu, G. Woodruff, Y. Wang, L. Graham, M. Hunt, D. Wu, E. Boehle, R. Ahmad, G. Poplawski, J. Brock, L.S.B. Goldstein, M.H. Tuszynski, Long-distance axonal growth from human induced pluripotent stem cells after spinal cord injury, Neuron 83 (2014) 789–796. https://doi.org/10.1016/j.neuron.2014.07.014.
[26] S. Tashiro, S. Nishimura, H. Iwai, K. Sugai, L. Zhang, M. Shinozaki, A. Iwanami, Y. Toyama, M. Liu, H. Okano, M. Nakamura, Functional Recovery from Neural Stem/Progenitor Cell Transplantation Combined with Treadmill Training in Mice with Chronic Spinal Cord Injury, Sci Rep 6 (2016) 30898. https://doi.org/10.1038/srep30898.
[27] P. Lu, S. Ceto, Y. Wang, L. Graham, D. Wu, H. Kumamaru, E. Staufenberg, M.H. Tuszynski, Prolonged human neural stem cell maturation supports recovery in injured rodent CNS, J Clin Invest 127 (2017) 3287–3299. https://doi.org/10.1172/JCI92955.
[28] Q. Ke, L. Li, B. Cai, C. Liu, Y. Yang, Y. Gao, W. Huang, X. Yuan, T. Wang, Q. Zhang, A.L. Harris, L. Tao, A.P. Xiang, Connexin 43 is involved in the generation of human-induced pluripotent stem cells, Hum Mol Genet 22 (2013) 2221–2233. https://doi.org/10.1093/hmg/ddt074.
[29] S. Kang, X. Chen, S. Gong, P. Yu, S. Yau, Z. Su, L. Zhou, J. Yu, G. Pan, L. Shi, Characteristic analyses of a neural differentiation model from iPSC-derived neuron according to morphology, physiology, and global gene expression pattern, Sci Rep 7 (2017) 12233. https://doi.org/10.1038/s41598-017-12452-x.
[30] A.S. Artyuhov, E.B. Dashinimaev, N.V. Mescheryakova, A.A. Ashikhmina, E.A. Vorotelyak, A.V. Vasiliev, Detection of small numbers of iPSCs in different heterogeneous cell mixtures with highly sensitive droplet digital PCR, Mol Biol Rep 46 (2019) 6675–6683. https://doi.org/10.1007/s11033-019-05100-2.
[31] X. Zeng, Y. Zeng, Y. Ma, L. Lu, B. Du, W. Zhang, Y. Li, W.Y. Chan, Bone marrow mesenchymal stem cells in a three-dimensional gelatin sponge scaffold attenuate inflammation, promote angiogenesis, and reduce cavity formation in experimental spinal cord injury, Cell Transplant 20 (2011) 1881–1899. https://doi.org/10.3727/096368911X566181.
[32] D.M. Basso, M.S. Beattie, J.C. Bresnahan, A sensitive and reliable locomotor rating scale for open field testing in rats, J Neurotrauma 12 (1995) 1–21. https://doi.org/10.1089/neu.1995.12.1.
[33] A. Ramón-Cueto, M.I. Cordero, F.F. Santos-Benito, J. Avila, Functional recovery of paraplegic rats and motor axon regeneration in their spinal cords by olfactory ensheathing glia, Neuron 25 (2000) 425–435.
https://doi.org/10.1016/s0896-6273(00)80905-8.
[34] B.-Q. Lai, X. Zeng, W.-T. Han, M.-T. Che, Y. Ding, G. Li, Y.-S. Zeng, Stem cell-derived neuronal relay strategies and functional electrical stimulation for treatment of spinal cord injury, Biomaterials 279 (2021) 121211. https://doi.org/10.1016/j.biomaterials.2021.121211.
[35] J. Wang, S. Sun, H. Deng, Chemical reprogramming for cell fate manipulation: Methods, applications, and perspectives, Cell Stem Cell 30 (2023) 1130–1147. https://doi.org/10.1016/j.stem.2023.08.001.
[36] K. Okita, T. Ichisaka, S. Yamanaka, Generation of germline-competent induced pluripotent stem cells, Nature 448 (2007) 313–317. https://doi.org/10.1038/nature05934.
[37] K. Okita, M. Nakagawa, H. Hyenjong, T. Ichisaka, S. Yamanaka, Generation of mouse induced pluripotent stem cells without viral vectors, Science 322 (2008) 949–953. https://doi.org/10.1126/science.1164270.
[38] International Stem Cell Initiative, K. Amps, P.W. Andrews, G. Anyfantis, L. Armstrong, S. Avery, H. Baharvand, J. Baker, D. Baker, M.B. Munoz, S. Beil, N. Benvenisty, D. Ben-Yosef, J.-C. Biancotti, A. Bosman, R.M. Brena, D. Brison, G. Caisander, M.V. Camarasa, J. Chen, E. Chiao, Y.M. Choi, A.B.H. Choo, D. Collins, A. Colman, J.M. Crook, G.Q. Daley, A. Dalton, P.A. De Sousa, C. Denning, J. Downie, P. Dvorak, K.D. Montgomery, A. Feki, A. Ford, V. Fox, A.M. Fraga, T. Frumkin, L. Ge, P.J. Gokhale, T. Golan-Lev, H. Gourabi, M. Gropp, G. Lu, A. Hampl, K. Harron, L. Healy, W. Herath, F. Holm, O. Hovatta, J. Hyllner, M.S. Inamdar, A.K. Irwanto, T. Ishii, M. Jaconi, Y. Jin, S. Kimber, S. Kiselev, B.B. Knowles, O. Kopper, V. Kukharenko, A. Kuliev, M.A. Lagarkova, P.W. Laird, M. Lako, A.L. Laslett, N. Lavon, D.R. Lee, J.E. Lee, C. Li, L.S. Lim, T.E. Ludwig, Y. Ma, E. Maltby, I. Mateizel, Y. Mayshar, M. Mileikovsky, S.L. Minger, T. Miyazaki, S.Y. Moon, H. Moore, C. Mummery, A. Nagy, N. Nakatsuji, K. Narwani, S.K.W. Oh, S.K. Oh, C. Olson, T. Otonkoski, F. Pan, I.-H. Park, S. Pells, M.F. Pera, L.V. Pereira, O. Qi, G.S. Raj, B. Reubinoff, A. Robins, P. Robson, J. Rossant, G.H. Salekdeh, T.C. Schulz, K. Sermon, J. Sheik Mohamed, H. Shen, E. Sherrer, K. Sidhu, S. Sivarajah, H. Skottman, C. Spits, G.N. Stacey, R. Strehl, N. Strelchenko, H. Suemori, B. Sun, R. Suuronen, K. Takahashi, T. Tuuri, P. Venu, Y. Verlinsky, D. Ward-van Oostwaard, D.J. Weisenberger, Y. Wu, S. Yamanaka, L. Young, Q. Zhou, Screening ethnically diverse human embryonic stem cells identifies a chromosome 20 minimal amplicon conferring growth advantage, Nat Biotechnol 29 (2011) 1132– 1144. https://doi.org/10.1038/nbt.2051.
[39] F.J. Rouhani, S. Nik-Zainal, A. Wuster, Y. Li, N. Conte, H. Koike-Yusa, N. Kumasaka, L. Vallier, K. Yusa, A. Bradley, Mutational History of a Human Cell Lineage from Somatic to Induced Pluripotent Stem Cells, PLoS Genet 12 (2016) e1005932. https://doi.org/10.1371/journal.pgen.1005932.
[40] S. Wang, Y. Du, B. Zhang, G. Meng, Z. Liu, S.Y. Liew, R. Liang, Z. Zhang, X. Cai, S. Wu, W. Gao, D. Zhuang, J. Zou, H. Huang, M. Wang, X. Wang, X. Wang, T. Liang, T. Liu, J. Gu, N. Liu, Y. Wei, X. Ding, Y. Pu, Y. Zhan, Y. Luo, P. Sun, S. Xie, J. Yang, Y. Weng, C. Zhou, Z. Wang, S. Wang, H. Deng, Z. Shen, Transplantation of chemically induced pluripotent stem-cell-derived islets under abdominal anterior rectus sheath in a type 1 diabetes patient, Cell 187 (2024) 6152-6164.e18. https://doi.org/10.1016/j.cell.2024.09.004.
[41] K. Kojima, H. Miyoshi, N. Nagoshi, J. Kohyama, G. Itakura, S. Kawabata, M. Ozaki, T. Iida, K. Sugai, S. Ito, R. Fukuzawa, K. Yasutake, F. Renault-Mihara, S. Shibata, M. Matsumoto, M. Nakamura, H. Okano, Selective Ablation of Tumorigenic Cells Following Human Induced Pluripotent Stem Cell-Derived Neural Stem/Progenitor Cell Transplantation in Spinal Cord Injury, Stem Cells Transl Med 8 (2019) 260–270. https://doi.org/10.1002/sctm.18-0096.
[42] M. Mandai, A. Watanabe, Y. Kurimoto, Y. Hirami, C. Morinaga, T. Daimon, M. Fujihara, H. Akimaru, N. Sakai, Y. Shibata, M. Terada, Y. Nomiya, S. Tanishima, M. Nakamura, H. Kamao, S. Sugita, A. Onishi, T. Ito, K. Fujita, S. Kawamata, M.J. Go, C. Shinohara, K.-I. Hata, M. Sawada, M. Yamamoto, S. Ohta, Y. Ohara, K. Yoshida, J. Kuwahara, Y. Kitano, N. Amano, M. Umekage, F. Kitaoka, A. Tanaka, C. Okada, N. Takasu, S. Ogawa, S. Yamanaka, M. Takahashi, Autologous Induced Stem-Cell-Derived Retinal Cells for Macular Degeneration, N Engl J Med 376 (2017) 1038–1046. https://doi.org/10.1056/NEJMoa1608368.
[43] M.W. Tibbitt, K.S. Anseth, Dynamic microenvironments: the fourth dimension, Sci Transl Med 4 (2012) 160ps24. https://doi.org/10.1126/scitranslmed.3004804.
[44] R. Yokoi, T. Shigemoto-Kuroda, N. Matsuda, A. Odawara, I. Suzuki, Electrophysiological responses to seizurogenic compounds dependent on E/I balance in human iPSC-derived cortical neural networks, J Pharmacol Sci 148 (2022)
[45] M.G. Boza-Morán, R. Martínez-Hernández, S. Bernal, K. Wanisch, E. Also-Rallo, A. Le Heron, L. Alías, C. Denis, M. Girard, J.-K. Yee, E.F. Tizzano, R.J. Yáñez-Muñoz, Decay in survival motor neuron and plastin 3 levels during differentiation of iPSC-derived human motor neurons, Sci Rep 5 (2015) 11696. https://doi.org/10.1038/srep11696.
[46] K. Satkunendrarajah, S.K. Karadimas, A.M. Laliberte, G. Montandon, M.G. Fehlings, Cervical excitatory neurons sustain breathing after spinal cord injury, Nature 562 (2018) 419–422. https://doi.org/10.1038/s41586-018-0595-z.
[47] C. Kathe, M.A. Skinnider, T.H. Hutson, N. Regazzi, M. Gautier, R. Demesmaeker, S. Komi, S. Ceto, N.D. James, N. Cho, L. Baud, K. Galan, K.J.E. Matson, A. Rowald, K. Kim, R. Wang, K. Minassian, J.O. Prior, L. Asboth, Q. Barraud, S.P. Lacour, A.J. Levine, F. Wagner, J. Bloch, J.W. Squair, G. Courtine, The neurons that restore walking after paralysis, Nature 611 (2022) 540–547. https://doi.org/10.1038/s41586-022-05385-7.
[48] V.N. Jensen, E.E. Huffman, F.L. Jalufka, A.L. Pritchard, S. Baumgartner, I. Walling, H. C Gibbs, D.A. McCreedy, W.J. Alilain, S.A. Crone, V2a neurons restore diaphragm function in mice following spinal cord injury, Proc Natl Acad Sci U S A 121 (2024) e2313594121. https://doi.org/10.1073/pnas.2313594121.
[49] T.H. Qazi, M.R. Blatchley, M.D. Davidson, F.M. Yavitt, M.E. Cooke, K.S. Anseth, J.A. Burdick, Programming hydrogels to probe spatiotemporal cell biology, Cell Stem Cell 29 (2022) 678–691. https://doi.org/10.1016/j.stem.2022.03.013.
[50] D. Crow, Could iPSCs Enable “Off-the-Shelf” Cell Therapy?, Cell 177 (2019) 1667– 1669. https://doi.org/10.1016/j.cell.2019.05.043.
[51] K.M. Piltti, G.M. Funes, S.N. Avakian, A.A. Salibian, K.I. Huang, K. Carta, N. Kamei, L.A. Flanagan, E.S. Monuki, N. Uchida, B.J. Cummings, A.J. Anderson, Increasing Human Neural Stem Cell Transplantation Dose Alters Oligodendroglial and Neuronal Differentiation after Spinal Cord Injury, Stem Cell Reports 8 (2017) 1534–1548. https://doi.org/10.1016/j.stemcr.2017.04.009.
[52] Q.L. Cao, Y.P. Zhang, R.M. Howard, W.M. Walters, P. Tsoulfas, S.R. Whittemore, Pluripotent stem cells engrafted into the normal or lesioned adult rat spinal cord are restricted to a glial lineage, Exp Neurol 167 (2001) 48–58. https://doi.org/10.1006/exnr.2000.7536.
[53] B.V. Lien, M.H. Tuszynski, P. Lu, Astrocytes migrate from human neural stem cell grafts and functionally integrate into the injured rat spinal cord, Exp Neurol 314 (2019) 46–57. https://doi.org/10.1016/j.expneurol.2019.01.006.
[54] C. Pavan, K.C. Davidson, N. Payne, S. Frausin, C.P.J. Hunt, N. Moriarty, M.Á. Berrocal Rubio, Z. Elahi, A.T. Quattrocchi, K.D. Abu-Bonsrah, L. Wang, W. Clow, H. Yang, M. Pellegrini, C.A. Wells, L.H. Thompson, A. Nagy, C.L. Parish, A cloaked human stem-cell-derived neural graft capable of functional integration and immune evasion in rodent models, Cell Stem Cell 32 (2025) 710-726.e8. https://doi.org/10.1016/j.stem.2025.03.008.
[55] M.S. Chen, A.B. Huber, M.E. van der Haar, M. Frank, L. Schnell, A.A. Spillmann, F. Christ, M.E. Schwab, Nogo-A is a myelin-associated neurite outgrowth inhibitor and an antigen for monoclonal antibody IN-1, Nature 403 (2000) 434–439.
https://doi.org/10.1038/35000219.
[56] G. Mukhopadhyay, P. Doherty, F.S. Walsh, P.R. Crocker, M.T. Filbin, A novel role for myelin-associated glycoprotein as an inhibitor of axonal regeneration, Neuron 13 (1994) 757–767. https://doi.org/10.1016/0896-6273(94)90042-6.
[57] K.C. Wang, V. Koprivica, J.A. Kim, R. Sivasankaran, Y. Guo, R.L. Neve, Z. He, Oligodendrocyte-myelin glycoprotein is a Nogo receptor ligand that inhibits neurite outgrowth, Nature 417 (2002) 941–944. https://doi.org/10.1038/nature00867.
[58] G.H.D. Poplawski, R. Lie, M. Hunt, H. Kumamaru, R. Kawaguchi, P. Lu, M.K.E. Schäfer, G. Woodruff, J. Robinson, P. Canete, J.N. Dulin, C.G. Geoffroy, L. Menzel, B. Zheng, G. Coppola, M.H. Tuszynski, Adult rat myelin enhances axonal outgrowth from neural stem cells, Sci Transl Med 10 (2018) eaal2563. https://doi.org/10.1126/scitranslmed.aal2563.
[59] G.H.D. Poplawski, R. Kawaguchi, E. Van Niekerk, P. Lu, N. Mehta, P. Canete, R. Lie, I. Dragatsis, J.M. Meves, B. Zheng, G. Coppola, M.H. Tuszynski, Injured adult neurons regress to an embryonic transcriptional growth state, Nature 581 (2020) 77–82. https://doi.org/10.1038/s41586-020-2200-5.
[60] Y. Li, X. He, R. Kawaguchi, Y. Zhang, Q. Wang, A. Monavarfeshani, Z. Yang, B. Chen, Z. Shi, H. Meng, S. Zhou, J. Zhu, A. Jacobi, V. Swarup, P.G. Popovich, D.H. Geschwind, Z. He, Microglia-organized scar-free spinal cord repair in neonatal mice, Nature 587 (2020) 613–618. https://doi.org/10.1038/s41586-020-2795-6.