¶ 《Journal of Neural Engineering》
¶ Organotypic human brain slice cultures as a translational testing platform for novel neuromodulation devices
Franz A M Eggert1,2,3,4, Berkhan Genc1,2, Sena Nur Arduc2, Anouk Wolters1,2, Kim Rijkers1,2,5,6, Kristen Kozielski7, Yasin Temel1,2,8 and Ali Jahanshahi1,2,9,∗
1 Department of Neurosurgery, Maastricht University Medical Center+, Maastricht, The Netherlands
2 Institute for Mental Health and Neuroscience, Maastricht University, Maastricht, The Netherlands
3 Department of Neurosurgery, Brandenburg Medical School, Campus Brandenburg, Brandenburg a. d. Havel, Germany
4 Faculty of Health Sciences, Joint Faculty of the Brandenburg University of Technology, Brandenburg Medical School and University of Potsdam, Potsdam, Germany
5 Centre for Integrative Neuroscience, Maastricht University, Maastricht, The Netherlands
6 Academic Center for Epileptology, Maastricht University Medical Center+, Maastricht, The Netherlands
7 Munich Institute of Biomedical Engineering, Technical University of Munich, Garching, Germany
8 Istanbul Atlas University, Istanbul, Turkey
9 Royal Netherlands Academy of Arts and Sciences, Netherlands Institute for Neuroscience, Amsterdam, The Netherlands
∗ Author to whom any correspondence should be addressed.
E-mail: a.jahanshahi@maastrichtuniversity.nl
¶ Keywords:
magneto-electric nanoparticles, neurostimulation, organotypic human brain slice cultures, translational model
¶ Abstract
Objective. To establish organotypic human brain slice cultures (hBSCs) as a translational screening platform for evaluating novel neuromodulation devices and to demonstrate the feasibility of the model using magnetoelectric nanoparticles (MENPs) as a representative neurostimulation modality. Approach. Viable hBSCs were prepared from resected cortical tissue of epilepsy surgery patients and GCaMP-based calcium imaging, multi-electrode array recordings, and immunohistochemical staining for c-Fos were conducted. The MENPs were injected into the hBSCs and stimulated with an alternating magnetic field to assess their neuromodulatory effects. Main Results. GCaMP transduction enables the real-time visualization of MENP-induced neuronal activity. Electrophysiological signals, including spiking and local field potentials, were observed in fresh, but not cultured, slices. c-Fos immunostaining revealed a significant increase in c-Fos expression in stimulated MENP-injected cultures compared to sham-treated controls. This protocol yielded reproducible tissue viability and consistent results across patient-derived samples. Significance. This technical note demonstrates that hBSCs represent a reproducible and ethically preferable translational model suitable for screening applications in neurotechnology research. The platform enables early-stage functional evaluation of neuromodulatory devices, particularly those with a higher risk of failure in vivo or curiosity-driven early-phase concepts in a setting superior to traditional in vitro approaches. This platform may help reduce reliance on animal models in neurotechnology development.
¶ 1. Introduction
With 9237 542 animals used for scientific purposes in 2022 in EU member states and Norway (EU 2022), the reliance of the scientific community on animal models cannot be underestimated (Keifer and Summers 2016). This reliance also holds for neuroscience, as it relies heavily on animal models, particularly rodents, to investigate complex brain functions and behavior (Keifer and Summers 2016,Moulin et al 2021). From an ethical perspective, the use of animals in scientific research can be justified (Machan 2002). However, according to the 4R’s framework (Reduction, Refinement, Replacement, and Responsibility) the capacity of animals to experience pain and suffering continues to present an ethical conflict in their use (Kiani et al 2022).
Several in vitro and ex vivo methodologies have been proposed as alternative platforms for testing of neural implants, particularly as screening tools for assessing tissue responses before progressing to in vivo studies (Gulino et al 2019). Conventional cell culture models, including human-induced pluripotent stem cells and immortalized cell lines, have been identified as valuable tools for central nervous systemrelated testing platforms (Nikolakopoulou et al 2020). However, they lack the structural and cellular complexity of native brain tissue. Brain-on-a-chip systems and organoids offer advanced alternatives; however, they still face challenges in replicating the intricate cytoarchitecture and functional connectivity of the human brain (Cassotta et al 2022).
Nanomaterials have been tested in organotypic brain slice cultures (BSCs) across different species, demonstrating their utility in studying neuroinflammation, tissue responses, and neurodegeneration (Kristensen et al 2001, Huuskonen et al 2005, Ereifej et al 2013, Usmani et al 2016). Despite these advancements, widely accepted testing tools for assessing nanomaterial toxicity in neural implants remain scarce (Chen et al 2020). Human BSCs (hBSCs) are advantageous because they preserve the human-specific cytoarchitecture, cellular diversity, extracellular matrix composition, and neuronal connectivity (Noraberg 2004, Schwarz et al 2019, Barth et al 2021). These attributes make hBSCs a compelling model for evaluating novel nanomaterials for neuromodulation, particularly during preliminary screening (Gulino et al 2019). This work introduces hBSCs as a translational testing platform for evaluating novel implantable devices for deep-brain neuromodulation. In the early stages of neurostimulation material development, various screening studies are required to assess stimulation parameters and material biocompatibility before progressing to in vivo experiments.
As magnetoelectric nanoparticles (MENPs) have already been tested in rodents, showing their potential to change neurophysiological activity and behavior (Kozielski et al 2021, Kumari et al 2024, DominguezParedes et al 2025), this study establishes a pipeline for hBSC-based testing of MENPs, validating tissue viability, and employing techniques such as calcium imaging, electrophysiology, and immunohistochemistry (IHC) for c-Fos to assess their potential for neuromodulation research in a reverse validation approach. c-Fos was selected as a marker of neuronal activity because of its well-established and widespread use in neuroscience research, and straightforward quantification (Lara Aparicio et al 2022).
¶ 2. Method
¶ 2.1. Brain tissue and cerebrospinal fluid collection
Eleven patients undergoing anterior temporal lobectomy (maximal temporal lobe resection in one case) for drug-resistant epilepsy between January 2023 and September 2024 at the Department of Neurosurgery of the Maastricht University Medical Center + (MUMC+) were asked to provide consent (METC 2024-0344) to collect resected brain tissue and human cerebrospinal fluid (hCSF) during their presurgical visit to the outpatient clinic.
After obtaining written informed consent, resected brain tissue was collected in the operating theatre immediately after resection. Furthermore, the hCSF removed by the surgeon as part of the standard procedure was collected in a tube and placed on ice (figure 1(A)). As part of general anesthesia, drugs such as propofol, norepinephrine, sufentanil, and dexamethasone were administered at typical doses. Following anterior temporal lobectomy, 1 × 1.5 cm brain tissue samples were taken from the posterior lateral region of the excised temporal neocortex specimen. The remaining tissue was used for histopathological analysis as part of standard medical process.
¶ 2.2. Magneto-electric nanoparticles
The MENPs consist of magnetostrictive CoFe2O4 nanoparticles coated with piezoelectric BaTiO3. When exposed to a direct (DC) magnetic field overlaid with an alternating (AC) field, the magnetostrictive core is deformed and strain is applied to the piezoelectric coating, thus eliciting an electric polarization. MENPs contained 36.1 0.6% BaTiO3 and 63.9 ± 0.6% CoFe2O4, and the average diameter was 277 18 nm with a zeta potential of 6.7 0.5 mV in artificial cerebrospinal fluid (aCSF) as measured in detail in a previous study of our group (Kozielski et al 2021). Before application, the MENPs were diluted in Milli-Q water (100 µg ml−1) and vortexed thoroughly.
¶ 2.3. hBSCs preparation
The organotypic human brain slice protocol was adopted from a previous work by Bak et al 2024. In summary, the neocortical brain tissue samples were immediately submerged in ice-cold, carbogenated (95% O2 5% CO2) slicing-aCSF (s-aCSF; figure 1, A). s-aCSF was composed of 110 mM choline chloride, 26 mM NaHCO3, 1.25 mM Na2HPO4∗2H2O, 11.6 mM sodium ascorbate, 3.1 mM sodium pyruvate, 7 mM MgCl2, 0.5 mM CaCl2, 2.5 mM KCl, 10 mM glucose dissolved in MilliQ, for incubation 1% penicillin/ streptomycin/ amphotericin B was added. Neuronal activity was blocked by choline, which replaces sodium, to mitigate the expected slicing stress. hCSF was centrifuged (4 ◦C, 2000 g) and sterile filtered to remove blood and either stored at −80 ◦C or used immediately.
After cleaning and trimming (figure 1(B)), brain slices with an area of approximately 0.75 0.75 cm and a thickness of 300 µm were produced using a vibratome (Leica VT1200S, Wetzlar,Germany; figures 1© and (D)). Throughout the procedure, the tissue was submerged in ice-cold carbogenated s-aCSF. Afterwards, the brain slices were kept at 37 ◦C in an air–liquid interface, including an intermediate step of HEPES medium (23.5 ml DMEM/F-12, 23.5 ml Neurobasal medium, 1 ml B27 supplement, 0.5 ml N2 supplement, 0.5 mL GlutaMAX and 0.5 ml non-essential amino acids are mixed to yield 50 ml) for 60 min. For cultivation, slices were transferred to a well plate with semi-permeable (0.4 µm pore size) net inserts (Corning® Transwell® 24 mm Transwell with 0.4 µm pore polyester membrane insert, TC-treated) serving as an air–liquid interface of hCSF and aCSF in a 1:1 ratio (125 mM NaCl, 25 mM NaHCO3, 2.5 mM KCl, 1.25 mM NaH2PO4, 1 mM MgCl2∗6 H2O, 2 mM CaCl2, 25 mM glucose diluted in MilliQ) interface and incubated for at least 48 h (37 ◦C, CO2 5%, O2 95% in humidified environment; figure 1(D)).

Figure 1. (A) Brain tissue acquisition and transport in ice-cold, oxygenated s-aCSF. (B, C) Preparation of organotypic cultures: a trimmed block of brain tissue is glued to agarose and sectioned in a vibratome chamber under continuous perfusion with ice-cold, oxygenated s-aCSF. (D) Cultures placed on an air–liquid interface membrane insert. (E), (F) injection of MENPs into the hBSC. (G) Custom-built stimulation coil used for magnetic actuation, with highlighted stimulation chamber. (H) MENPinjected hBSC fixated beneath a custom resin net grid prior to immersion for calcium imaging to prevent floating. (I) ∗Custom made acrylic well holder, red arrows point at well holder within the stimulation chamber, white arrows indicate a gap between the coil and the microscope as to prevent any vibration being transmitted to the microscope. (J) Active calcium imaging set-up (acrylic well holder as seen in (I) now holding well/ hBSC/ resin net under stimulation and ongoing fluorescent microscopy). (K) Multi-electrode array (MEA) with a net-fixated MENP injected hBSC (∗(black) harp slice grid, ∗(white) hBSC with visible MENP aggregates, +MEA chip). (L) ∗Zoomed out MEA chamber with arrows pointing at CSF-perfusion system.
¶ 2.4. Viral transduction
The cultures were then transduced using adeno associated retrograde viral vector (AAV) derived from pGP-AAV-syn-jGCaMP7f-WPRE (Addgene, Massachusetts, USA). The AAV was applied by bringing a 2 µl drop into contact with the surface of the hBSC. Brain slices were cultured for 3, 7 and 10 d, during which 0.75 ml of aCSF was replaced in well plates every other day to optimize transduction efficiency. On the 10th day post-transduction, the experiments were conducted.
¶ 2.5. Stimulation and fluorescence image acquisition
hBSCs were randomly assigned into two groups: MENP and stimulation (M+/S+) and MENP and no stimulation (M+/S ). Both groups received 2 µl injections of MENP suspension (∼3 µg of MENPs), followed by incubation to allow MENP diffusion (figures 1(E) and (F)). After injection, the M+/S+ group was stimulated with a custom coil system (figure 1(G)) at 220 mT DC; 6 mT AC, 140 Hz. The stimulation parameters were based on our recent in vivo and in silico studies, through which we have developed and tested MENP-based neurostimulation technology (Kozielski et al 2021, Kumari et al 2024, Dominguez-Paredes et al 2025). The stimulation was performed under a Leica FS fixed-stage microscope (Leica Microsystems GmbH, Wetzlar, Germany) equipped with a fluorescence illumination system and a 20×/0.50 numerical aperture (NA) objective water-dipping lens (figures 1(H)–(J)). Slices in M+/S− group was handled in the same manner, but with the coil turned off such that the tissue was only exposed to the DC magnetic field. Throughout the calcium imaging data acquisition, hBSCs were submerged in carbogenated aCSF and mechanically stabilized using a custom-made net grid (figure 1(H)). Low-intensity fluorophore light and reduced exposure time were used to minimize phototoxicity and photobleaching. Vibration issues were addressed using linear stack alignment with SIFT in ImageJ software [version 1.8; National Institutes of Health, Bethesda, USA] to ensure precise image alignment. Additionally, background subtraction was performed to correct for photobleaching effects and enhance the accuracy and clarity of the resulting images. Two hours after calcium imaging, M+/S+ and M+/S− groups received DC/AC and DC stimulation respectively, followed by 2 h of incubation and fixation paraformaldehyde for cFos IHC.
¶ 2.6. Multi-electrode array recordings
For electrophysiological recordings with multielectrode array (MEA), aCSF was prepared by dissolving the following in 500 ml of Milli-Q water: 3.68 g NaCl (126 mM), 0.112 g KCl (3 mM), 1.008 g NaHCO₃ (24 mM), 0.991 g glucose (11 mM), 0.147 g CaCl₂ (2.65 mM), 0.154 g MgSO₄ (2.56 mM), and 0.078 g NaH₂PO₄ (1.3 mM). The pH of the solution was adjusted to 7.4. Brain slices were incubated in carbogenated aCSF for 1.5 h prior to recording, which was performed using the CorePlate™ SingleWell HD-MEAs, a high-density MEA system (3Brain AG, Switzerland; figures 1(K) and (L)). hBSCs were stimulated in the coil prior to recording.
¶ 2.7. Immunohistochemical staining
After fixation in paraformaldehyde for 24 h, the tissue slices were incubated in 20% sucrose for another 24 h, then snap-frozen, sectioned into 20 µm slices, and stored at −80 ◦C until staining. c-Fos staining was used to assess neuronal activity (Morgan and Curran 1986). Tissue sections were incubated overnight with polyclonal c-Fos (rabbit 1:1000; Santa Cruz Biotechnology Inc.; sc-253), followed by biotinylated donkey anti-rabbit secondary antibody (1:400; Jackson ImmunoResearch Laboratories Inc.; 711065152) and avidin–biotin peroxidase complex (1:800; Elite ABC kit, Vector Laboratories; PK-6100). Staining was visualized using 3,3′-diaminobenzidine combined with NiCl2 intensification.
¶ 2.8. Bright-field image acquisition and quantification of c-Fos positive neurons
Bright-field images were acquired using an Olympus AX 70 microscope (Olympus, Zoeterwoude, The Netherlands). With a 10 /0.30 NA objective and CellP software (version 6.1). The lamp intensity stabilized at 6.8 V, and the exposure time was set to 80 ms in the monochrome mode for all samples. A daily calibration of pixel-to-distance (1 pixel = 0.49 µm) was conducted using a stage micrometer. For each section, three non-overlapping 1 mm2 cortical regions were systematically imaged from the medial to the lateral edge. Image analysis was carried out using FIJI (ImageJ v1.54) by an evaluator blinded to the experimental grouping and conditions: Renyi entropy autothresholding followed by manual adjustment (±5%), ROI delineation, and multi-point counting of NiDAB-labeled c-Fos positive nuclei. Cell counts were normalized to area and averaged across 3–5 sections per slice, and statistical analysis was performed using GraphPad Prism 10.1.2 (GraphPad Software, San Diego, California, USA).
¶ 3. Results
¶ 3.1. Study population
Eleven experiments were conducted using neocortical tissue samples obtained from 11 patients who underwent anterior temporal lobectomy with amygdalohippocampectomy for epilepsy treatment at Maastricht University Medical Center between January 2023 and September 2024 (table 1).
¶ 3.2. Viral transduction and calcium imaging
During the study, viral transduction was halted on days 3, 7, and 10 to perform fluorescence imaging, with the optimal results observed on day 10 (figures 2(A)–(F)). Optimal’ refers to the combination of reproducible, robust GCaMP fluorescence, preserved neuronal morphology, and visible spontaneous calcium transients across multiple fields of view, indicating mature viral expression and functional neuronal activity suitable for subsequent stimulation experiments. hBSCs were submerged in aCSF and held in place using a 3D-printed net (figure 1(H)) to allow for accurate image acquisition. The coil had to be detached from the microscope to avoid vibration (figure 1(I)). The image was adjusted by carefully moving the custom-made hBSC-well holder (figure 1(J)). Fluorescence stacks were analyzed using Fiji (Schindelin et al 2012), where background subtraction was applied to mitigate the effects of photobleaching and temporal noise. To further stabilize the images and prevent flickering artifacts, the ‘Linear Stack Alignment with SIFT’ plugin was employed.
Table 1. Overview of surgical procedures and histopathological findings in patients undergoing anterior temporal lobectomy for epilepsy. The table summarizes the type and side of surgery performed and neuropathology of the neocortex.
| Exp | Age | Sex | Side | Histopathological analysis neocortex |
| 1 | 61 | M | Right | Minor gliotic changes |
| 2 | 55 | F | Left | Mild gliotic changes |
| 3 | 46 | F | Right | Minor gliotic changes in the temporal lobe,severe hippocampal sclerosis |
| 4 | 60 | M | Right | Minor hemorrhagic changes |
| 5 | 32 | M | Left | Minor gliotic changes |
| 6 | 41 | M | Left | Minor gliotic changes |
| 7 | 27 | F | Right | Minor gliotic changes |
| 8 | 32 | M | Left | Minor gliotic changes. |
| 9 | 44 | M | Left | Minor gliotic changes |
| 10 | 57 | F | Left | Minor gliotic changes |
| 11 | 39 | M | Left | Minor to moderate gliotic changes |
¶ 3.3. Multi electrode array recordings
MEA recordings were performed in a 1:1 mixture of hCSF and aCSF. Fresh, carbogenated aCSF was continuously perfused through the MEA chamber throughout the recordings (figure 1(L)). Because the magnetic field of the coil impaired the performance of the device, stimulation had to take place before placing the hBSC on the MEA. Notably, spikes and local field potentials (LFPs) were only detected in carbogenated aCSF supplemented with 7.5 mM KCl or 500 µM 4-aminopyridine. Recordings were attempted on cultured human brain slices immediately after preparation and on days 2, 5, and 7. However, LFPs and spikes were exclusively observed on the day of sectioning. Consequently, electrophysiological recordings were conducted immediately after slicing the brain tissue to a thickness of 300 µm prior to culturing.
¶ 3.4. IHC and c-Fos quantification
c-Fos expression was clearly detectable within the tissue (figures 3(A) and (B)) along with visible aggregates of MENPs (figure 3©). In addition to the intended nuclear staining, cytoplasmic c-Fos signals were observed; however, only cells with clearly distinguishable nuclear staining were included in the analysis. Although not consistently observed, group-dependent differences in c-Fos expression were at times discernible by qualitative assessment (figures 3(D)–(G)).
Pooled c-Fos counts from the respective experimental conditions were assessed for normality using the Shapiro–Wilk test, and the data violated assumptions of normality (p < 0.0001). c-Fos-positive cell counts were normalized within each experimental group, and group differences in fold change were subsequently analyzed using the non-parametric twotailed Mann–Whitney test. A significantly higher fold-change in c-Fos-positive cells was observed in the M+/S+ group than in the M+/S− group (p = <0.001) (figure 3(H)).
¶ 4. Discussion
This study aimed to evaluate whether hBSCs represent a translational testing platform for assessing the neuromodulatory potential of novel technologies using MENPs as a representative example. We successfully prepared hBSCs, performed viral transduction, and modulated neuronal activity by using MENPs. Neuronal modulation and culture viability were confirmed using a dual readout setup for calcium imaging and IHC staining for c-Fos. The visible expression of c-Fos in all experimental groups implied tissue viability at the time of fixation (Lara Aparicio et al 2022), whereas significant differences in expression levels between groups showed the ability of MENPs to modulate neuronal activity.
¶ 4.1. Customization of BSCs setup
Several adaptations were made to optimize the procedure for specific needs, improve reproducibility, and address the technical challenges encountered during implementation. The protocol described by Bak et al was both thorough and adaptable. Effective communication with the surgical team was crucial, and we found it essential to be present in the operation room prior to opening the dura mater to collect hCSF for incubation. When feasible, instead of suctioning, the surgeon aspirated excess hCSF using a syringe, particularly after accessing the subdural space, allowing immediate cooling on ice. Regarding tissue preparation, we removed the pia mater only when it was clearly identifiable or naturally detached, as manual dissection often caused cortical damage. Notably, vibratome sectioning was unaffected by its presence. For transferring tissue slices, a blunt spatula or probe minimized mechanical stress. After trimming and positioning hBSCs on net inserts, excess liquid was removed using a 1000 µl pipette. If hBSCs overlapped, folded, or were mispositioned, the precise application of small aCSF droplets resolved these issues. Depending on the amount of brain tissue obtained, we produced 18, 36, or 44 hBSCs, with each well containing three slices. In theory, MENP injection could occur at any time between production and at least 2 h prior to stimulation. However, immediate injection resulted in visible particle migration from the hBSCs to the net insert, which was reduced when injection was performed 2 h prior to stimulation. We tested two injection setups: a microinjector and a 2 µl pipette. While the microinjector allowed for precise spatial approximation, its static tubing system led to particle settling, clogging, or uneven distribution. The 10 µl pipette proved more practical, allowing agitation of the MENP suspension immediately before injection. We first formed a droplet at the pipette tip, brought it into contact with the tissue until the surface tension broke, and injected the remaining volume. The insert was removed from the plate and placed in a single well for stimulation. The wells were removed from the incubator for stimulation and returned. The experimental groups were assigned to the individual wells. All groups were handled identically to prevent detection of c-Fos expression due to mechanical stress.
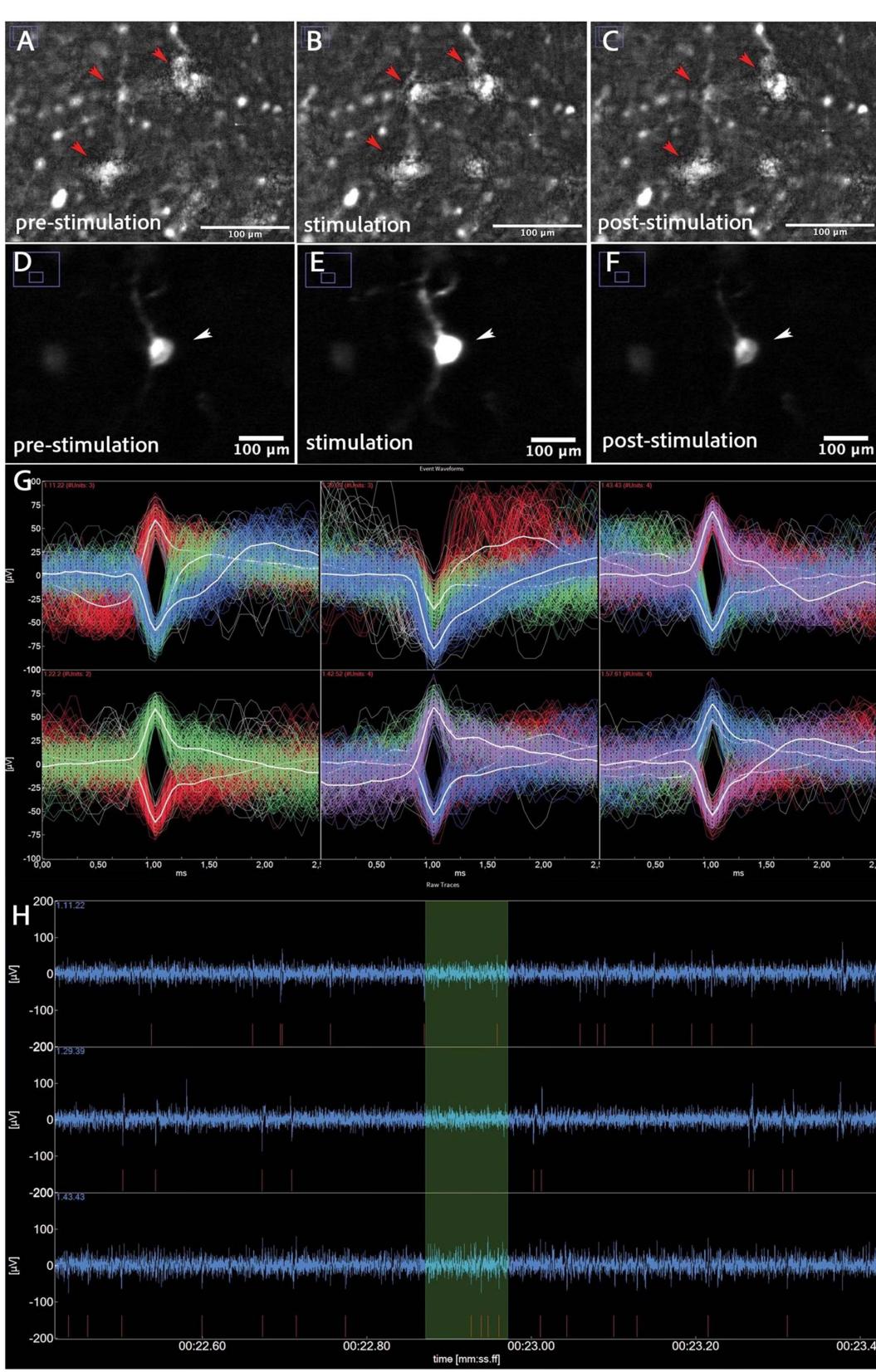
Figure 2. (A) GCaMP positive neurons pre-stimulation (B) GCaMP positive neurons showing network activity (arrows) whilst under stimulation. © GCaMP positive neurons, post-stimulation. (D) Single GCaMP positive neuron (arrow), pre-stimulation. (E) Single GCaMP positive neuron (arrow) showing calcium influx under stimulation. (F) Single GCaMP positive neuron (arrow) with reduced calcium influx, post-stimulation. (G) Spike waveforms. Each panel corresponds to a different electrode channel, displaying multiple event-aligned waveforms over a 2–3 ms window. The superimposed traces (in red, white, green, and blue) reflect individual neuronal spikes and/or population responses, demonstrating variability in amplitude and shape across electrodes. (H) Raw electrophysiological trace recording. The blue waveform shows extracellular voltage over time, reflecting spontaneous neuronal activity. Red markings beneath the trace indicate individual spike detection events (putative action potentials). Green shaded regions highlight intervals of interest (burst detection windows). The relatively stable baseline and discrete spike events suggest viable neuronal health and clear signal-to-noise separation.
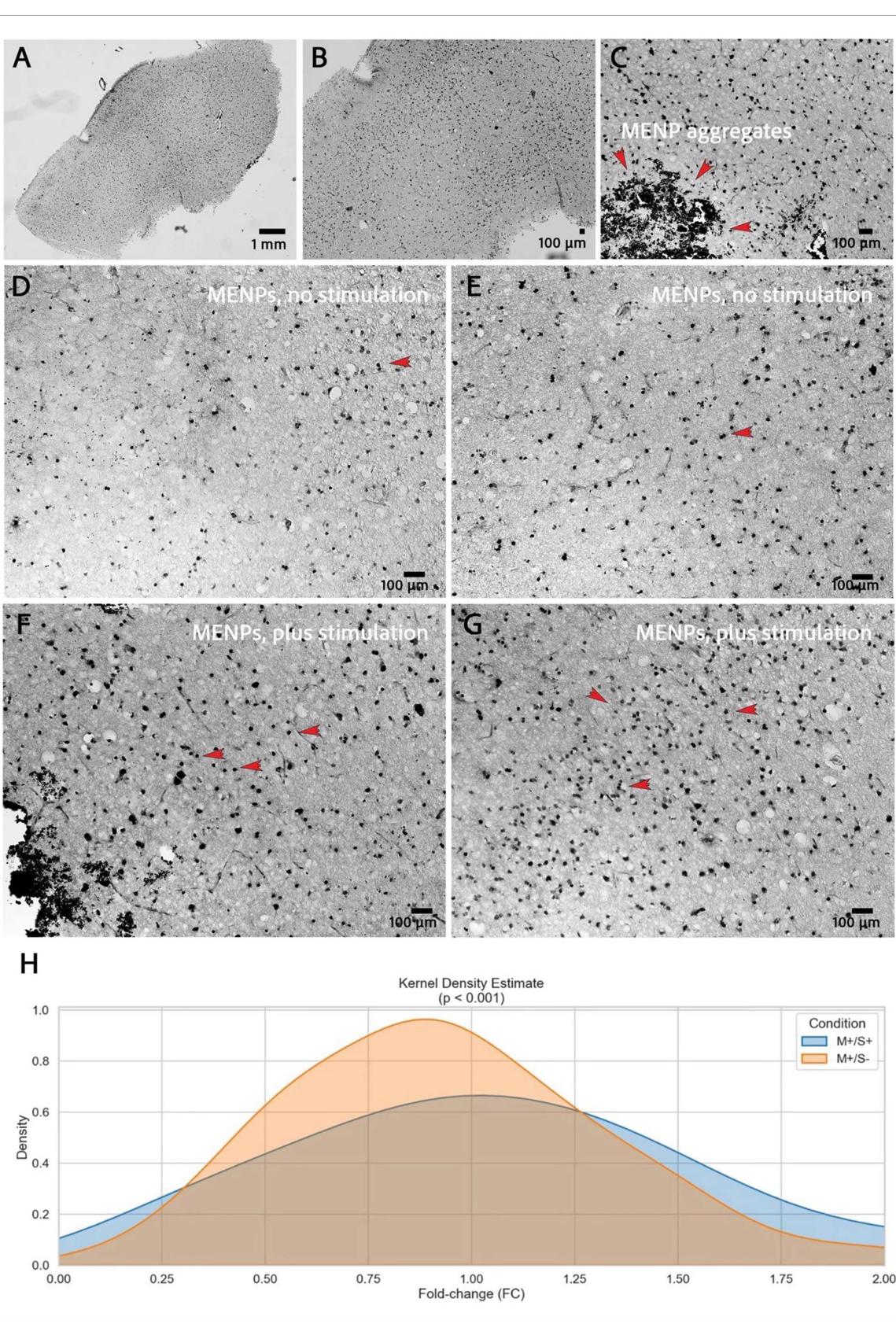
Figure 3. (A) Overview image of a whole hBSC-slice, stained for c-Fos. (B) Visible dense c-Fos expression. © Magnification used to count c-Fos positive nuclei and exemplary MENP aggregates. (D/E) IHC images of hBSCs from the MENP nostimulation group showing visibly less c-Fos expression (red arrows indicate exemplary c-Fos-positive nuclei). (F/G) IHC images of hBSCs from the MENP stimulation group showing visibly increased c-Fos expression density (red arrows indicate exemplary c-Fos-positive nuclei). (H) c-Fos expression of M+/S+ vs M+/S− groups fold change, visualized by Kernel Density estimate (Mann–Whitney p = <0.001).
The staining protocol largely followed standard procedures; however, special attention was required during tissue mounting because of fragility of the slices. We used a cut-off pipette to transfer the slices onto glass slides, removing excess liquid with tissue paper, and a 10 µl pipette. Following overnight drying, we recommend extending the oven incubation period from one to three hours to improve adhesion and prevent tissue detachment during dehydration.
¶ 4.2. Evaluation of hBSCs as a translational model
We aimed to determine whether hBSCs, as prepared and maintained in this system, can serve as a robust and scalable platform for functional studies and screening tools. This evaluation is central to our overarching motivation to reduce dependence on animal experiments by advancing human ex vivo models.
Depending on the experimental design, several cultures could have been produced for testing, for example, different stimulation regimes, particle loads, or readouts such as electrophysiological recordings. When applied to animal experiments, this would necessitate multiple ethical reviews and significant investments in time and money. Furthermore, as there are significant differences in gene expression and cell morphology between rodents and humans (Hodge et al 2019), the observability of any effects or mechanisms in hBSCs should be mandatory before proceeding to in vivo experiments, thus highlighting its capability as a translational tool for promising new approaches. A significant challenge when working with this model is the heterogeneity in patient characteristics, such as age, sex, medication (antiepileptic drugs that directly influence neuronal activity), peri-surgical conditions, and morphology of the retrieved tissue. Although this can be tackled by either including a multitude of cases and showing an overall significant difference, as we did, or by treating every experiment as standing alone and performing analysis only within hBSCs drawn from one case. The choice of readout method introduces additional challenges. For example, c-Fos is commonly used as a marker for neuronal activity because it is relatively easy to detect and is widely recognized for this purpose. However, these limitations should also be considered. c-Fos is not exclusively expressed by neurons but also by other central nervous system cells, such as glial cells. Furthermore, mechanical stimuli can induce c-Fos expression in glia and neurons (Jaworski et al 2018). These mechanical stimuli pose practical challenges because handling cultures during experiments exerts mechanical stress that may trigger c-Fos expression. It is crucial to handle cultures from different conditions as consistently as possible and to minimize false-positive detections by waiting at least two hours after injecting cultures with MENPs before assessing c-Fos expression. In contrast, electrical stimuli exclusively induce c-Fos expression in neurons (Hisanaga et al 1990, Cruz-Mendoza et al 2022). Additionally, cFos expression reaches its maximum earlier in astrocytes, within 20–45 min after stimulation (CruzMendoza et al 2022), whereas peak expression in neurons occurs at approximately two hours (Lara Aparicio et al 2022). Therefore, the differences quantified at the selected time point are expected to reflect neuronal activation in response to MENP stimulation. To further characterize the c-Fos-positive cells, double immunofluorescence labeling can be performed depending on the study design. Spontaneous epileptiform activity originating from the resected tissue may lead to false-positive electrophysiological recordings. Although this concern is not strongly supported by existing literature, it warrants acknowledgment. Spontaneous interictal or ictal activity at the single-cell level is a rare phenomenon (Levinson et al 2020). Notably, a study employing a similar MEA-based experimental setup, co-culturing an epileptic human neocortical biopsy with hiPSCderived cortical neurons, also demonstrated that epileptiform network activity emerged only in the hybrid co-culture, whereas neither the biopsy nor the neuronal layer alone showed seizure-like discharges (Hu et al 2023).
¶ 5. Conclusion
The hBSC model demonstrated the feasibility of studying novel neurostimulation devices in functional human neuronal networks. This approach provides an ethically advantageous and translational testing platform that can be used to pre-screen promising technologies before testing in animal studies, especially those with a higher risk of failure in vivo or curiosity-driven early-phase products. Identifying effective interventions in human tissue may reduce the number of required animal experiments while increasing the relevance and impact of subsequent in vivo research. As ethical and funding frameworks increasingly limit the use of animal models, human-based systems like hBSCs are wellpositioned to become essential tools in translational neuroscience. Future work will focus on additional experiments and refining the described readouts, further enhancing the platform for systematic evaluation of emerging nanoscale and bioelectronic neuromodulation technologies.
Written informed consent was obtained from all participants prior to inclusion in the study. Tissue collection was performed in compliance with the institutional and national guidelines for research involving human biological materials.
¶ Data availability statement
The data cannot be made publicly available upon publication because no suitable repository exists for hosting data in this field of study. The data that support the findings of this study are available upon reasonable request from the authors.
¶ Acknowledgments
This work was supported by a grant from the Michael J Fox Foundation (MJFF-019718) to AJ, KK and YT; ROMO (2023) to AJ, KR and YT, the European Union (ERC, NANeurO, Project Number 101115997) to KK, and the Netherlands research council (NWO-Vidi: 09150171910022) to AJ. FE was supported by the Thiemann Foundation through a doctoral scholarship. FE thanks Dr Faisal Alosaimi and Dr Ghazi Al Jowf for their support.
¶ Conflict of interest
Franz A M Eggert acknowledges the support from the Thiemann Foundation through a doctoral scholarship. The foundation had no role in the study design, data collection, analysis, decision to publish, or manuscript preparation. All other authors declare no competing interests.
¶ Ethics statement
Human brain tissue and cerebrospinal fluid (CSF) were obtained from patients undergoing anterior temporal lobectomy for drug-resistant epilepsy at the Maastricht University Medical Center (MUMC+). All procedures involving human participants were conducted in accordance with the Declaration of Helsinki and were approved by the local medical ethics committee (METC 2022-3539 and 2024-0344).
¶ Author contributions
Franz A M Eggert 0000-0003-0235-1720 Conceptualization (equal), Data curation (lead), Formal analysis (lead), Investigation (lead), Methodology (equal), Visualization (equal), Writing – original draft (lead), Writing – review & editing (lead)
Berkhan Genc 0000-0001-9578-6584 Conceptualization (equal), Data curation (equal), Formal analysis (equal), Investigation (equal), Methodology (equal), Software (lead), Validation (equal), Visualization (equal), Writing – review & editing (equal)
Sena Nur Arduc 0009-0003-3941-0576 Data curation (equal), Investigation (equal)
Anouk Wolters 0000-0001-6975-1812 Methodology (equal), Writing – review & editing (equal)
Kim Rijkers 0000-0002-3348-158X Conceptualization (equal), Investigation (equal), Project administration (equal), Resources (equal), Supervision (equal), Writing – review & editing (equal)
Kristen Kozielski Methodology (equal)
Yasin Temel 0000-0002-3589-5604 Funding acquisition (lead), Project administration (equal), Resources (equal)
Ali Jahanshahi 0000-0002-7451-3461 Conceptualization (equal), Funding acquisition (lead), Methodology (equal), Project administration (lead), Resources (lead), Supervision (lead), Writing – review & editing (equal)
¶ References
Bak A et al. 2024. Human organotypic brain slice cultures: a detailed and improved protocol for preparation and long-term maintenance. J. Neurosci. Methods, 404, 110055.
Barth M et al. 2021. Microglial inclusions and neurofilament light chain release follow neuronal alpha-synuclein lesions in long-term brain slice cultures. Mol. Neurodegener, 16, 54.
Cassotta M et al. 2022. The future of Parkinson’s disease research: a new paradigm of human-specific investigation is necessary… and possible. ALTEX, 39, 694–709.
Chen R J et al. 2020. The current understanding of autophagy in nanomaterial toxicity and its implementation in safety assessment-related alternative testing strategies. Int. J. Mol. Sci., 21, 2387.
Cruz-Mendoza F, Jauregui-Huerta F, Aguilar-Delgadillo A, Garcia-Estrada J, Luquin S. 2022. Immediate early gene c-Fos in the brain: focus on glial cells. Brain Sci., 12, 687.
Dominguez-Paredes D, Temel Y, Milojkovic A, Mulder-Jongen D, Kumari P, Ries M, Kozielski K, Jahanshahi A. 2025. Magnetoelectric nanoparticle technology for mitigating motor deficits in Parkinsonian mice. Brain Stimul., 18, 1551–1560.
Ereifej E S, Cheng M M, Mao G, VandeVord P J. 2013. Examining the inflammatory response to nanopatterned polydimethylsiloxane using organotypic brain slice methods. J. Neurosci. Methods, 217, 17–25.
EU. 2022. EU-wide animal research statistics. Available at: https://www.understandinganimalresearch.org.uk/news/eu-wideanimal-research-statistics-2022.
Gulino M, Kim D, Pane S, Santos S D, Pego A P. 2019. Tissue response to neural implants: the use of model systems toward new design solutions of implantable microelectrodes. Front. Neurosci., 13, 689.
Hisanaga K, Sagar S M, Hicks K J, Swanson R A, Sharp F R. 1990. c-Fos proto-oncogene expression in astrocytes associated with differentiation or proliferation but not depolarization. Brain Res. Mol. Brain Res., 8, 69–75.
Hodge R D et al. 2019. Conserved cell types with divergent features in human versus mouse cortex. Nature, 573, 61–68.
Hu M H Y, Frimat J-P, Rijkers K, Schijns O E M G, van den Maagdenberg A M J M, Dings J T A, Luttge R, Hoogland G. 2023. Spontaneous epileptic recordings from hiPSC-derived cortical neurons cultured with a human epileptic brain biopsy on a multi electrode array. Appl. Sci., 13, 1432.
Huuskonen J, Suuronen T, Miettinen R, van Groen T, Salminen A. 2005. A refined in vitro model to study inflammatory responses in organotypic membrane culture of postnatal rat hippocampal slices. J. Neuroinf., 2, 25.
Jaworski J, Kalita K, Knapska E. 2018. c-Fos and neuronal plasticity: the aftermath of Kaczmarek’s theory. Acta Neurobiol. Exp., 78, 287–296.
Keifer J, Summers C H. 2016. Putting the “biology” back into “neurobiology”: the strength of diversity in animal model systems for neuroscience research. Front. Syst. Neurosci., 10, 69.
Kiani A K et al. 2022. Ethical considerations regarding animal experimentation. J. Prev. Med. Hyg., 63, E255–E266.
Kozielski K L, Jahanshahi A, Gilbert H B, Yu Y, Erin O, Francisco D, Alosaimi F, Temel Y, Sitti M. 2021. Nonresonant powering of injectable nanoelectrodes enables wireless deep brain stimulation in freely moving mice. Sci. Adv., 7, eabc4189.
Kristensen B W, Noraberg J, Thiebaud P, Koudelka-Hep M, Zimmer J. 2001. Biocompatibility of silicon-based arrays of electrodes coupled to organotypic hippocampal brain slice cultures. Brain Res., 896, 1–17.
Kumari P, Wunderlich H, Milojkovic A, Lopez J E, Fossati A, Jahanshahi A, Kozielski K. 2024. Multiscale modeling of magnetoelectric nanoparticles for the analysis of spatially selective neural stimulation. Adv. Healthcare Mater., 13, e2302871.
Lara Aparicio S Y et al. 2022. Current opinion on the use of c-Fos in neuroscience. NeuroSci, 3, 687–702.
Levinson S, Tran C H, Barry J, Viker B, Levine M S, Vinters H V, Mathern G W, Cepeda C. 2020. Paroxysmal discharges in tissue slices from pediatric epilepsy surgery patients: critical role of GABA(B) receptors in the generation of ictal activity. Front. Cell Neurosci., 14, 54.
Machan T R. 2002. Why human beings may use animals. J. Value Inq., 36, 9–14.
Morgan J I, Curran T. 1986. Role of ion flux in the control of c-Fos expression. Nature, 322, 552–555.
Moulin T C, Covill L E, Itskov P M, Williams M J, Schioth H B. 2021. Rodent and fly models in behavioral neuroscience: an evaluation of methodological advances, comparative research, and future perspectives. Neurosci. Biobehav. Rev., 120, 1–12.
Nikolakopoulou P, Rauti R, Voulgaris D, Shlomy I, Maoz B M, Herland A. 2020. Recent progress in translational engineered in vitro models of the central nervous system. Brain, 143, 3181–3213.
Noraberg J. 2004. Organotypic brain slice cultures: an efficient and reliable method for neurotoxicological screening and mechanistic studies. Altern Lab Animal, 32, 329–337.
Schindelin J et al. 2012. Fiji: an open-source platform for biological-image analysis. Nat. Methods, 9, 676–682.
Schwarz N et al. 2019. Long-term adult human brain slice cultures as a model system to study human CNS circuitry and disease. eLife, 8, e48417.
Usmani S et al. 2016. 3D meshes of carbon nanotubes guide functional reconnection of segregated spinal explants. Sci. Adv., 2, e1600087.